Advertisements
Advertisements
प्रश्न
Write the first four members of the homologous series that begins with H-C≡C-H. Also, write down their general molecular formula.
Advertisements
उत्तर
| No. | Name of a compound and molecular formula | Structural formula |
| 1. | Ethyne (C2H2) | HC ≡ CH |
| 2. | Propyne (C3H4) | CH3 - C ≡ CH |
| 3. | But-1-yne (C4H6) | CH3 - CH2 - C ≡ CH |
| 4. | Pent-1-yne (C5H8) | CH3 - CH2 - CH2 - C ≡ CH |
Comparing these molecular formulae and assigning the number of carbon atoms as ‘n’, the following general formula is deduced: CnH2n - 2 (where n = 2, 3, 4...)
APPEARS IN
संबंधित प्रश्न
Identify the functional group in the following compound.
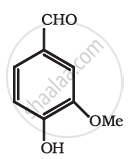
Find out all the functional groups present in the following polyfunctional compound.
Thyroxine, the principal thyroid hormone.

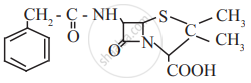
Find out all the functional groups present in the following polyfunctional compound.
Penicillin G, a naturally occurring antibiotic.
Identify primary, secondary, tertiary, and quaternary carbon in the following compound.
\[\begin{array}{cc}\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\|\phantom{.....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
How many cyclic and acyclic isomers are possible for the molecular formula C3H6O?
Write a note on homologous series.
Write the molecular formula of the first six members of homologous series of nitro – alkanes.
What is the percentage of carbon in urea?
(Atomic mass C = 12, H = 1, N = 14, O = 16)
An organic compound containing carbon, hydrogen and oxygen is soluble in dil. H2SO4 and does not react with sodium metal or KMnO4. When it is heated with HI in excess, a single alkyl halide is obtained. The original compound can be ____________.
Which of the following is a hetero-aromatic benzenoid compound?
How many 3° carbon atoms are present in 1, 4 dibromo-2-methylpentane?
How many primary, secondary and tertiary carbon atoms respectively are present in isobutane?
An organic compound 'X' (molecular formula C6H7O2N) has six carbons in a ring system, two double bonds and also a nitro group as a substituent, 'X' is ______.
The correct decreasing order of priority for the functional groups of organic compounds in the IUPAC system of nomenclature is ______.
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{...............}\\
\ce{CH3}\phantom{..}\ce{CH3}\phantom{.............}
\end{array}\]
Identify the α - carbons in the following species and give the total number of
\[\ce{CH3 -CH2 -\overset{⊕}{C}H -CH2 -CH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary, and quaternary carbons in the following compound.
\[\begin{array}{cc}
\ce{CH3\phantom{.................}}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3 CH3\phantom{.............}}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H - CH2 - CH3}\]
Indicate the σ and π bonds in the following molecule:
\[\ce{HCONHCH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}\\
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
