Advertisements
Advertisements
प्रश्न
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
पर्याय
ΔU > 0, W > 0
ΔU < 0, W > 0
ΔU < 0, W < 0
ΔU = 0, W > 0
Advertisements
उत्तर
ΔU < 0, W > 0
APPEARS IN
संबंधित प्रश्न
Give an example of some familiar process in which heat is added to an object, without changing its temperature.
Give an expression for work done in an isothermal process.
Give the equation of state for an adiabatic process.
Draw the PV diagram for the isochoric process.
Derive the work done in an isothermal process.
Explain in detail the isochoric process.
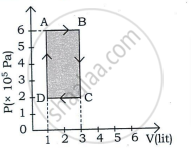
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______

Assertion: Equal volumes of monatomic and polyatomic gases are adiabatically compressed separately to equal compression ratio `("P"_2/"P"_1)`. Then monatomic gas will have greater final volume.
Reason: Among ideal gases, molecules of a monatomic gas have the smallest number of degrees of freedom.
Give any two types of a thermodynamic process.
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.