Advertisements
Advertisements
प्रश्न
The total number of tetrahedral voids in the face-centered unit cell is ______.
पर्याय
6
8
10
12
Advertisements
उत्तर
The total number of tetrahedral voids in the face-centered unit cell is 8.
Explanation:
Fee unit cell contains 8 tetrahedral voids at centre of each 8 smaller cube of a unit cell as shown below
Eight tetrahedral voids per fee unit vell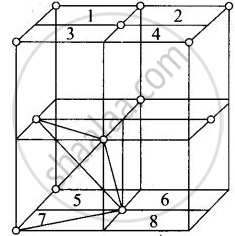
Each cube represented by numeric 1, 2, 3, 4, 5, 6, 7, 8 contains one tetrahedral void.

Each cube contains one tetrahedral void its body centre as shown above

APPEARS IN
संबंधित प्रश्न
Which crystal system has axial angles equal to 90°?
Calculate the number of unit cells present in 1 g of gold which has a face-centered cubic lattice.
Coordination numbers of Zn2+ and S2– in the crystal structure of wurtzite are ____________.
In face-centered cubic lattice, a unit cell is shared equally by how many unit cells?
In zinc blend structure:
A unit cell of BaCl2 (fluorite structure) is made up of ____________.
How many lithium atoms are present in a unit cell with edge length 3.5 Å and density 0.53 g cm−3? (Atomic mass of Li = 6.94):
Assertion: Total number of octahedral voids present in unit cell of cubic close packing including the one that is present at the body centre, is four.
Reason: Besides the body centre there is one octahedral void present at the centre of each of the six faces of the unit cell and each of which is shared between two adjacent unit cells.
CsBr, crystallises in a body centred cubic lattice. The unit cell edge length is 436.6 pm. Given that the atomic mass of Cs = 133 a. m. u and that of Br = 80 a. m. u and Avogadro's number being 6.02 × 1023 mol–1, the density of Cs Br is:- .
Na and Mg crystallise in Bee and Fcc type crystals respectively. "The no. of atoms in the unit cells of their respective crystals is:
Sodium crystallises in a body-centred cubic unit cell. (bcc) with edge length 4.29 A. what is the radius of the sodium atom? What is the length of the body diagonal of the unit cell?
The packing fraction for a body-centred cube is
The density of a crystal is given by the formula
The correct option for the number of body-centered unit cells in all 14 types of Bravais lattice unit cells is ______
Orthorhombic crystal has the following unit cell dimensions ______.
A certain element crystallises in a bcc lattice of unit cell edge length 27 A°. If the same element under the same conditions crystallises in the fcc lattice, the edge length of the unit cell in A° will be ______. (Round off to the Nearest Integer).
[Assume each lattice point has a single atom]
Gold crystallizes in a face-centered cubic lattice. If the length of the edge of the unit cell is 407 pm. The density of gold assuming it to be spherical is ______ g/cm3. Atomic mass of gold = 197 amu.
Which primitive unit cell has unequal edge lengths (a ≠ b ≠ c) and all axial angles different from 90°?
