Advertisements
Advertisements
प्रश्न
The total number of tetrahedral voids in the face-centered unit cell is ______.
विकल्प
6
8
10
12
Advertisements
उत्तर
The total number of tetrahedral voids in the face-centered unit cell is 8.
Explanation:
Fee unit cell contains 8 tetrahedral voids at centre of each 8 smaller cube of a unit cell as shown below
Eight tetrahedral voids per fee unit vell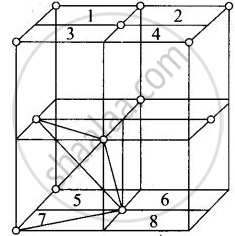
Each cube represented by numeric 1, 2, 3, 4, 5, 6, 7, 8 contains one tetrahedral void.

Each cube contains one tetrahedral void its body centre as shown above

APPEARS IN
संबंधित प्रश्न
Gold (atomic radius = 0.144 nm) crystallises in a face-centred unit cell. What is the length of a side of the cell?
The space lattice of graphite is ____________.
Coordination numbers of Zn2+ and S2– in the crystal structure of wurtzite are ____________.
In face-centered cubic lattice, a unit cell is shared equally by how many unit cells?
In zinc blend structure:
A unit cell of BaCl2 (fluorite structure) is made up of ____________.
The distance between Na– and Cl– ions in NaCl with a density 2.165 g cm–3 is __________.
A crystal has the lattice parameters a ≠ b ≠ c and α = β = γ = 90°. The crystal system is
CsBr, crystallises in a body centred cubic lattice. The unit cell edge length is 436.6 pm. Given that the atomic mass of Cs = 133 a. m. u and that of Br = 80 a. m. u and Avogadro's number being 6.02 × 1023 mol–1, the density of Cs Br is:- .
Na and Mg crystallise in Bee and Fcc type crystals respectively. "The no. of atoms in the unit cells of their respective crystals is:
How many kind of space lattice are possible in a crystal.
The density of a crystal is given by the formula
A certain element crystallises in a bcc lattice of unit cell edge length 27 A°. If the same element under the same conditions crystallises in the fcc lattice, the edge length of the unit cell in A° will be ______. (Round off to the Nearest Integer).
[Assume each lattice point has a single atom]
Gold crystallizes in a face-centered cubic lattice. If the length of the edge of the unit cell is 407 pm. The density of gold assuming it to be spherical is ______ g/cm3. Atomic mass of gold = 197 amu.
Which primitive unit cell has unequal edge lengths (a ≠ b ≠ c) and all axial angles different from 90°?
