Advertisements
Advertisements
प्रश्न
The structural formula of an ester is :

Write the formula of the acid and the alcohol from which it is formed.
Advertisements
उत्तर
he structural formula of the ester corresponds to the compound ethylethanoate (CH3COOC2H5). Esters are sweet smelling substances which are formed when an alcohol reacts with carboxylic acid in the presence of concentrated sulphuric acid.
(a) The formula of the ethanoic acid is CH3COOH.
(b) The formula of the alcohol (ethanol) is CH3-CH2-OH.
The chemical equation showing the reactants and the products is as follows:
`CH_3 COOH + CH_3 CH_2 OH` 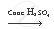 `CH_3 COOCH_2 CH_3 + H_2 O`
`CH_3 COOCH_2 CH_3 + H_2 O`
APPEARS IN
संबंधित प्रश्न
Identify the term or substance based on the descriptions given below
Ice like crystals formed on cooling an organic acid sufficiently.
Give the common names and IUPAC names of the following compounds of CH3COOH.
How does ethanoic acid react with sodium hydrogen carbonate? Give equation of the reaction which takes place.
Give any two uses of ethanoic acid.
Consider the following organic compounds:
HCHO, C2H5OH, C2H6, CH3COOH, C2H5CI
Choose two compounds which can react in the presence of conc. H2SO4 to form an ester. Give the name and formula of the ester formed.
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is a constituent of vinegar?(e) Which compound is a constituent of vinegar?
How is acetic acid prepared from acetylene?
Give the structural formula of Acetic acid.
How is ethyne prepared in the laboratory?
Why is pure acetic acid also known as glacial acetic acid?
Choose the correct answer:
Select the acid which contains four hydrozen atoms in it.
State how the following conversions can be carried out:
Ethene to Ethyl alcohol.
Write the characteristics of ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Draw the structural diagram of:
Ethanoic acid
Ethanoic acid reacts with the basic salt, sodium carbonate, to form a salt, named ______, water and carbon dioxide gas.
What happens when a small piece of sodium is dropped in ethanol? Write the equation for this reaction.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
