Advertisements
Advertisements
प्रश्न
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
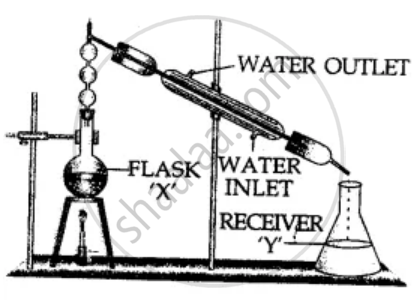
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
Advertisements
उत्तर
-
No, they can be separated by separating funnel.
- The physical property on which separation is based “Difference in their boiling points.”
-
Water having boiling point 100°C will remain in flask ‘X’.
- Alcohol having lower B.P. 78°C will evaporate first and condense in the conical flask — receiver ‘Y’.
- The upper part of the ‘Fractionating column is cooler, so as the hot vapours rise up in the column, they get cooled (condense) and trickle back into the distillation flask ‘X’.
APPEARS IN
संबंधित प्रश्न
Fill in the blank
Use of a ................. to separate the components of a mixture of solids is based on the difference in the size of the components
State the number of atoms of each element, present in \[\ce{HNO3}\].
Also name the compound.
State the number of atoms of CaCO3
Name: Three different forms of carbon
Indicate whether the following statement is true or false:
A compound is made up of just one kind of atom
Indicate whether the following statement is true or false:
Noble gases are high reactive
Explain the difference between Metals and non-metals.
Select the correct answer to the statement given below:
The chemical name for dinitrogen oxide [N20].
Differentiate between homogenous & heterogeneous mixtures.
What is the Latin name of Sodium?
