Advertisements
Advertisements
प्रश्न
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
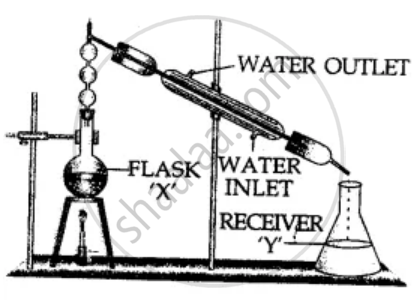
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
Advertisements
उत्तर
-
No, they can be separated by separating funnel.
- The physical property on which separation is based “Difference in their boiling points.”
-
Water having boiling point 100°C will remain in flask ‘X’.
- Alcohol having lower B.P. 78°C will evaporate first and condense in the conical flask — receiver ‘Y’.
- The upper part of the ‘Fractionating column is cooler, so as the hot vapours rise up in the column, they get cooled (condense) and trickle back into the distillation flask ‘X’.
APPEARS IN
संबंधित प्रश्न
Name the substance you will add to speed up sedimentation.
Define: Molecule
Indicate whether the following statement is true or false:
All elements are artificially prepared
Tick (√) the correct alternative from the choice given for the following statement.
All pure substances have
Tick (√) the correct alternative from the choice given for the following statement.
Gold is used to make jewellery because
Complete the statement given below by filling in the blank with the correct word.
From the elements nitrogen, chlorine, bromine, the element present in the atmosphere is ________.
Write the names of elements from the following symbol:
Ti
‘An atom is the basic unit of an element’. Draw a diagram of an atom – divisible as seen today.
Select the correct answer from A, B, C, D & E for the statement given below :
A metalloid.
Match the following.
| 1. | Element | Made up of molecules |
| 2. | Compound | Impure substance |
| 3. | Colloid | Settles down on standing |
| 4. | Suspension | Pure substance |
| 5. | Mixture | Made up of atoms |
