Advertisements
Advertisements
प्रश्न
Sodium hydroxide solution is added to solution A. A white precipitate is formed which is soluble in excess sodium hydroxide. Name the metal ion present in A.
Advertisements
उत्तर
The metal ion present in solution A is Pb2+.
APPEARS IN
संबंधित प्रश्न
What do you observe when caustic soda solution is added to the following solution, first a little and then in excess:
FeCl3
Write a balanced equation for this reaction.
Using sodium hydroxide solution, how will you distinguish: Zinc nitrate solution from calcium nitrate solution
Using sodium hydroxide solution, how will you distinguish:
Iron (II) chloride from iron (III) chloride
State what do you observe when:
Neutral litmus solution is added to alkaline solution
Sodium hydroxide solution is added to the solutions containing the ions mentioned in List X. List Y gives the details of the precipitate. Match the ions with their coloured precipitates.
| List X | List Y |
| (i) Pb2+ | (A) Reddish Brown |
| (ii) Fe2+ | (B) White insoluble inexcess |
| (iii) Zn2+ | (C) Dirty green |
| (iv) Fe3+ | (D) White soluble in excess |
| (v) Cu2+ | (E) White soluble in excess |
| (vi) Ca2+ | (F) Blue |
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to lead nitrate solution and zinc nitrate solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to ferrous nitrate and lead nitrate.
What is observed when hot, concentrated caustic soda solution is added to zinc? Write a balanced equation.
Aqueous lead (II) nitrate can be distinguished from aqueous zinc nitrate by adding any of the following solutions in excess, except:
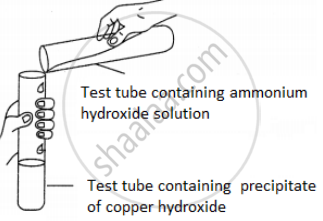
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.