Advertisements
Advertisements
प्रश्न
Show on a graph the variation of the de Broglie wavelength (λ) associated with an electron, with the square root of accelerating potential (V) ?
Advertisements
उत्तर
`lambda = 1.22/sqrtV`
`therefore lambda sqrtV=` const
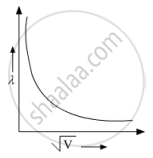
Hence, we get a hyperbola.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
