Advertisements
Advertisements
प्रश्न
Propanal and pentan-3-one are the ozonolysis products of an alkene? What is the structural formula of the alkene?
Advertisements
उत्तर
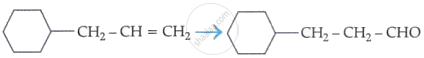
As per the given information, propanal and pentan-3-one are the ozonolysis products of an alkene. Let the given alkene be ‘A’. Writing the reverse of the ozonolysis reaction, we get:

The products are obtained on the cleavage of ozonide ‘X’. Hence, ‘X’ contains both products in the cyclic form. The possible structure of ozonide can be represented as:
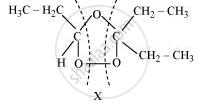
Now, ‘X’ is an addition product of alkene ‘A’ with ozone. Therefore, the possible structure of alkene ‘A’ is:
\[\begin{array}{cc}
\ce{H3C - CH2 - CH = C - CH2 - CH3}\\
\phantom{.......}|\\
\phantom{..............}\ce{CH2CH3}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
An alkene ‘A’ on ozonolysis gives a mixture of ethanal and pentan-3-one. Write structure and IUPAC name of ‘A’.
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Write a chemical equation for combustion reaction of the following hydrocarbon:
Butane
Write a chemical equation for combustion reaction of the following hydrocarbon:
Pentene
Write a chemical equation for combustion reaction of the following hydrocarbon:
Hexyne
The addition of HBr to 1-butene gives a mixture of products A, B and C
| (A) | 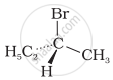 |
| (B) |  |
| (C) | CH3 – CH2 – CH2 – CH2 – Br |
The mixture consists of:
Which of the following alkenes on ozonolysis give a mixture of ketones only?
| (i) | CH3 – CH = CH – CH3 |
| (ii) | \[\begin{array}{cc} \ce{CH3 - C - CH = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | 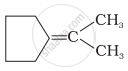 |
| (iv) | \[\begin{array}{cc} \phantom{...................}\ce{CH3}\\ \phantom{..............}/\\ \ce{(CH3)2 C = C}\\ \phantom{..............}\backslash\\ \phantom{...................}\ce{CH3} \end{array}\] |
An alkene 'X' on ozonolysis produces two moles of isovaleraldehyde. Predict the IUPAC name of the alkene.
Which of the following reagent is used for the following reaction?
\[\ce{CH3CH2CH3 ->[?] CH3CH2CHO}\]
The major product formed in the following reactions is:
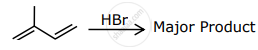

Select schemes A, B, C out of
(I) acid catalysed hydration
(II) HBO
(III) oxymercuration-demercuration
What would be the main product when propene reacts with HBr?
Propene, \[\ce{CH3 - CH = CH2}\] can be converted to 1-propanol by oxidation. Which set of reagents among the following is ideal to effect the conversion ______.
3-Methyl-pent-2-ene on reaction with HBr in presence of peroxide forms an addition product. The number of possible stereoisomers for the product is ______.
In the presence of peroxide, HCl and HI do not give anti-Markovnikov's addition of alkenes because ______.
An alkene ‘A’ contains three C-C, eight C-H σ bonds and one C-C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write the IUPAC name of ‘A’.
Identify the correct reagents that would bring about the following transformation.