Advertisements
Advertisements
प्रश्न
Match the detergents given in Column I with their uses given in Column II.
| Column I | Column II |
(i) 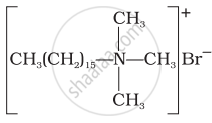 |
(a) Dishwashing powder |
(ii) 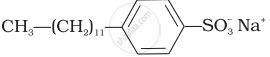 |
(b) Laundry soap |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (c) Hair conditioners |
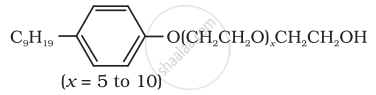
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (d) Toothpaste |
Advertisements
उत्तर
| Column I | Column II |
(i)  |
(c) Hair conditioners |
(ii)  |
(d) Toothpaste |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (b) Laundry soap |
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (a) Dishwashing powder |
Explanation:
(i) Hair shampoos/conditioners are made up of cationic detergents. These are quaternary ammonium salts of amines with chlorides, bromides or acetates, e.g., cetyltrimethyl ammonium bromide.
(ii) Anionic detergents are used in toothpaste e.g., sodium dodecylbenzene sulphonate. It can be prepared as follows.
\[\ce{\underset{Lauryl alcohol}{CH3(CH2)10CH2OH} ->[H2SO4] \underset{Laury hydrogensulphate}{CH3(CH2)10CH2OSO3H} ->[NaOH(aq)] \underset{(Anionic detergent)}{\underset{Sodium laurylsulphate}{CH3(CH2)10CH2OS\overset{-}{O}\overset{+}{N}a}}}\]

(iii) Laundry soaps contain fillers like sodium rosinate. Sodium silicate, borax and sodium carbonate. Sodium rosinate makes the soap to lather well.
(iv) Dishwashing powder are non-ionic detergents.