Advertisements
Advertisements
प्रश्न
In which of the following molecules carbon atom marked with asterisk (*) is asymmetric?
(a) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(b) \[\begin{array}{cc}
\ce{D}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(c) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{OH}\phantom{..}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{H}\phantom{...}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
पर्याय
(a), (b), (c), (d)
(a), (b), (c)
(b), (c), (d)
(a), (c), (d)
Advertisements
उत्तर
(a), (b), (c)
Explanation:
Asymmetric/chiral carbon atom is that in which all of its four valencies with four different groups or atoms.
In molecules (a), (b) and (c), all have asymmetric carbon as each carbon has satisfied all four valencies with four different groups or atoms.
In molecule (d), carbon satisfies two of its valencies with two hydrogen atoms i.e., similar atom. So, it is not an asymmetric carbon atom.
APPEARS IN
संबंधित प्रश्न
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)3CCH2CH(Br)C6H5
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Draw the structure of major monohalo product in each of the following reactions :
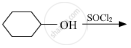
Which is the correct IUPAC name for \[\begin{array}{cc}
\ce{CH3 - CH - CH2 - Br}\\
\phantom{}|\phantom{.......}\\
\phantom{}\ce{C2H5}\phantom{....}
\end{array}\]?
What should be the correct IUPAC name for diethylbromomethane?
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(a) Aryl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (b) Alkyl halide |
| (iii) | 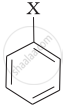 |
(c) Vinyl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (d) Allyl halide |
Why are haloalkanes more reactive towards nucleophilic substitution reactions than haloarenes and vinylic halides?
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH2CH2C(CH3)2CH2I}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(CI)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Which of the following are secondary bromides?
