Advertisements
Advertisements
प्रश्न
Identify the salts P and Q from the observation given below:
On performing the flame test salt P produces a lilac coloured flame and its solution gives a white precipitate with silver nitrate solution. Which is soluble in ammonium hydroxide solution.
Advertisements
उत्तर
KCl
APPEARS IN
संबंधित प्रश्न
Why is water not added to concentrated H2SO4 in order to dilute it?
Why is H2SO4 is not used as a drying agent for H2S?
What would you observe in the following case?
Concentrated sulphuric acid is added to equal volumes of cold water.
Give reason for the following:
When carbon is heated with concentrated sulphuric acid, carbon dioxide is formed.
Give reason for the following:
H2SO4 cannot be prepared by heating sodium sulphate with conc. HCl or HNO3
Give examples of the use of sulphuric acid as
An oxidizing agent
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Zinc hydroxide
What do you see when concentrated sulphuric acid is added to copper sulphate-5-water ( You are not required to say what is happing, nor is it necessary to name the products.)
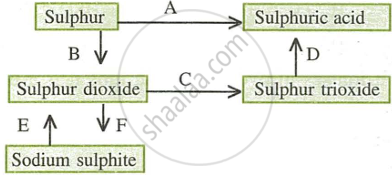
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Give one equation to show the following property of sulphuric acid:
Dehydrating property
