Advertisements
Advertisements
प्रश्न
Identify the gas evolved and give the chemical test in the following case:
Dilute hydrochloric acid reacts with iron (II) sulphide.
Advertisements
उत्तर
Gas Evolved: When dilute hydrochloric acid reacts with iron (II) sulphide, the gas evolved is hydrogen sulphide.
Reaction:
\[\ce{FeS + 2HCl -> FeCl2 + H2S ^}\]
Chemical Test for Hydrogen Sulphide: To test for H2S gas, you can use lead acetate paper:
- Moisten a strip of lead acetate paper.
- Expose it to the gas that evolved.
Observation: If H2S is present, the lead acetate paper will turn black due to the formation of lead sulphide (PbS).
\[\ce{Pb(C2H3O2)2 + H2S -> PbS + 2CH3COOH}\]
संबंधित प्रश्न
Name the products formed when hot and concentrated sulphuric acid reacts with Copper.
Give reason for the following:
Concentrated sulphuric acid is kept in airtight bottles.
The following statement is correct only under certain conditions. Rewrite the statement including the appropriate conditions.
Oxalic acid reacts with sulphuric acid to produce carbon monoxide and carbon dioxide.
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Zinc hydroxide
In this question, you required to supply the word (or words) that will make the sentence correct. Rewrite the copper statement.
Copper sulphate crystals are dehydrate by sulphuric acid.
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Sodium Sulphate from sodium carbonate.
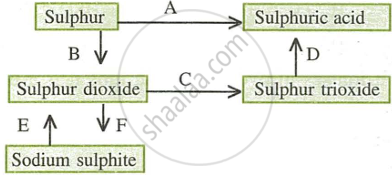
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Write the equation of the following reaction :
Concentrated sulphuric acid is poured over sugar
Write a balanced equation for the following conversion:
Lead sulphate from lead nitrate and sulphuric acid.
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
State the method of collection of the gas formed above.
