Advertisements
Advertisements
प्रश्न
Find the maximum Coulomb force that can act on the electron due to the nucleus in a hydrogen atom.
Advertisements
उत्तर
Charge on the electron, q1 = `1.6 xx 10^-19 C`
Charge on the nucleus, q2 = `1.6 xx 10^-19 C`
Let r be the distance between the nucleus and the electron.
Coulomb force (F) is given by
`F = (q_1q_2)/(4 pi∈_0r^2) ........(1)`
Here , q1 = q2 = q = 1.6`xx 10^-19C`
000 Smallest distance between the nucleus and the first orbit, r = 0.53 `r = 0.53xx10^-10m `
`K= 1/(4piepsilon_0) = 9 xx 10^9Nm^2C^-2`
Substituting the respective values in (1), we get
`F =((9xx10^9)xx(1.6xx10^-19)xx(1.6xx10^-19))/(0.53xx10^-10)^2`
= `(1.6xx1.6xx9xx10^-9)/(0.53) = 82.02 xx 10^-9`
= `8.2xx10^-8 N`
APPEARS IN
संबंधित प्रश्न
A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. What series of wavelengths will be emitted?
Find the wavelength of the electron orbiting in the first excited state in hydrogen atom.
The first excited energy of a He+ ion is the same as the ground state energy of hydrogen. Is it always true that one of the energies of any hydrogen-like ion will be the same as the ground state energy of a hydrogen atom?
What will be the energy corresponding to the first excited state of a hydrogen atom if the potential energy of the atom is taken to be 10 eV when the electron is widely separated from the proton? Can we still write En = E1/n2, or rn = a0 n2?
The minimum orbital angular momentum of the electron in a hydrogen atom is
Which of the following curves may represent the speed of the electron in a hydrogen atom as a function of trincipal quantum number n?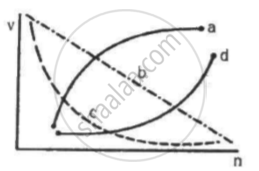
As one considers orbits with higher values of n in a hydrogen atom, the electric potential energy of the atom
Which of the following products in a hydrogen atom are independent of the principal quantum number n? The symbols have their usual meanings.
(a) vn
(b) Er
(c) En
(d) vr
Ionization energy of a hydrogen-like ion A is greater than that of another hydrogen-like ion B. Let r, u, E and L represent the radius of the orbit, speed of the electron, energy of the atom and orbital angular momentum of the electron respectively. In ground state
Find the binding energy of a hydrogen atom in the state n = 2.
A hydrogen atom emits ultraviolet radiation of wavelength 102.5 nm. What are the quantum numbers of the states involved in the transition?
(a) Find the first excitation potential of He+ ion. (b) Find the ionization potential of Li++ion.
A hydrogen atom in state n = 6 makes two successive transitions and reaches the ground state. In the first transition a photon of 1.13 eV is emitted. (a) Find the energy of the photon emitted in the second transition (b) What is the value of n in the intermediate state?
A gas of hydrogen-like ions is prepared in a particular excited state A. It emits photons having wavelength equal to the wavelength of the first line of the Lyman series together with photons of five other wavelengths. Identify the gas and find the principal quantum number of the state A.
Suppose, in certain conditions only those transitions are allowed to hydrogen atoms in which the principal quantum number n changes by 2. (a) Find the smallest wavelength emitted by hydrogen. (b) List the wavelength emitted by hydrogen in the visible range (380 nm to 780 nm).
Consider an excited hydrogen atom in state n moving with a velocity υ(ν<<c). It emits a photon in the direction of its motion and changes its state to a lower state m. Apply momentum and energy conservation principles to calculate the frequency ν of the emitted radiation. Compare this with the frequency ν0 emitted if the atom were at rest.
Let En = `(-1)/(8ε_0^2) (me^4)/(n^2h^2)` be the energy of the nth level of H-atom. If all the H-atoms are in the ground state and radiation of frequency (E2 - E1)/h falls on it ______.
- it will not be absorbed at all.
- some of atoms will move to the first excited state.
- all atoms will be excited to the n = 2 state.
- no atoms will make a transition to the n = 3 state.
A hydrogen atom makes a transition from n = 5 to n = 1 orbit. The wavelength of photon emitted is λ. The wavelength of photon emitted when it makes a transition from n = 5 to n = 2 orbit is ______.
