Advertisements
Advertisements
प्रश्न
Explain the following
A glass bottle completely filled with water and tightly closed at room temperature is likely to burst when kept in the freezer of a refrigerator.
Advertisements
उत्तर
Inside the freezer, when the temperature of water falls below 4°C, the water in the bottle starts expanding. If the bottle is completely filled and tightly closed, there is no space for water to expand, and hence, the bottle may burst.
APPEARS IN
संबंधित प्रश्न
Water is used as a cooling agent.
What is the role of the anomalous behaviour of water in preserving aquatic life in regions of cold climate?
Draw a graph to show the variation in density of water with temperature in the range from 0°C to 10°C.
Density of water is maximum at :
Explain, why does a glass bottle completely filled with water and tightly capped burst when placed in a freezer?
The following diagrams illustrate three situations involving thermometers which are labeled A, Band C. In each situation the thermometers indicate different readings.
(i) What do you expect the approximate reading of the thermometer B and C would be? Give a reason for your answer.
(ii) How would the readings of A and B help you in calibrating a thermometer?
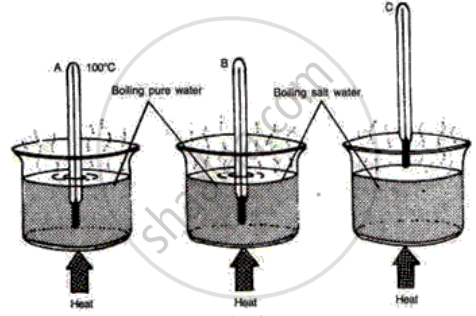
Study the following diagrams and write down your observations.

In a region with a cold climate the aquatic animals can survive at 4 °C, because _______.
