Advertisements
Advertisements
प्रश्न
Draw a resonance structure of the following:
Buta-1,3-diene
Advertisements
उत्तर
Resonance structures of buta-1,3-diene:

APPEARS IN
संबंधित प्रश्न
Find out the most stable species from the following. Justify
`dot"CH","CH"_3-dot"CH" - "CH"_3,` \[\begin{array}{cc}\ce{CH3 -\dot{C} - CH3}\\
|\phantom{.}\\\phantom{..}\ce{CH3}\end{array}\]
Draw a resonance structure of the following:
Phenol
Write true or false. Correct the false statement.
Homolytic fission involves the unsymmetrical breaking of a covalent bond.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Draw all the no-bond resonance structures of isopropyl carbocation.
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
The correct IUPAC name of the compound 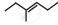 is ______.
is ______.
Choose the correct option.
The geometry of a carbocation is ______.
Predict the order of reactivity of the following compounds by SNl reaction mechanism.
\[\ce{\underset{\text{(I)}}{C6H5CH(C6H5)Cl}}\]
\[\ce{\underset{\text{(II)}}{C6H5CH2Cl}}\]
\[\ce{\underset{\text{(III)}}{C6H5C(CH3)(C6H5)Cl}}\]
Which among the following is a set of nucleophiles?
Which of the following is TRUE for homolytic fission?
The most unstable free radical among the following is:
The overlap of σ-p orbitals is called ____________.
Which of the following statements is not correct?
The most probable product in the reaction given below is:

Identify the functional group that has an electron-donating inductive effect.
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogens.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Which of the following groups exhibits a +I effect?
