Advertisements
Advertisements
प्रश्न
Distinguish the properties of solid, liquid, and gas. Draw a suitable diagram.
Advertisements
उत्तर
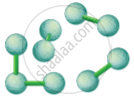
| S.No. | Solid | Liquid | Gas |
| 1. | Definite shape and volume | No definite shape, it attains the shape of the vessel | it has neither a definite shape nor a definite volume. |
| 2. | Incompressible | Compressible to a small extent | Highly compressible. |
| 3. | Little space between the particles | Greater space between the particles | Greater space between the particles. |
| 4. | Particles cannot move freely | Particles move freely | Particles are in a continuous, random motion. |
| 5. |
Particles in a Solid
Eg. Stone |
Particles in a Liquid
Eg. Water |
Particles in a Gas
Eg. Air |
APPEARS IN
संबंधित प्रश्न
Explain why, the diffusion of a solid in another solid is a very slow process.
Give reasons
A gas fills up the space available to it.
The three main states of matter are solids, liquids, and gases. Compare the three states with reference to the following characteristics of matter
(a) volume
(b) shape
(c) compressibility
(d) diffusion.
Matter in any state is composed of particles. Compare the three states of matter i,e. solids, liquids, and gases with reference to:
(a) intermolecular space
(b) the intermolecular force of attraction
(c) movement of particles
Explain the term ‘Interconversion of matter’. With reference to ice, water, and water vapour show diagrammatically the change of state of matter from solid to liquid to gaseous and back to the original state.
Identify the state of matter based on the arrangement of the molecules.
The force of attraction between liquid particles is more than solid particles.
Complete the Analogy.
Bucket: Plastic:: Table: ______.
What is the physical state of water at 100ºC?
The density of solid Argon is 1.6 ml at - 233°C. If the Argon atom is assumed to be sphere of radius 1.5 × 10-8 cm, then the ______ % of solid Argon is apparently occupied.
[Take : NA = 6 × 1023, Atomic mass of Ar = 40]