Advertisements
Advertisements
प्रश्न
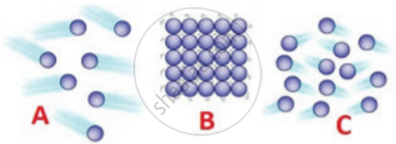
Matter in any state is composed of particles. Compare the three states of matter i,e. solids, liquids, and gases with reference to:
(a) intermolecular space
(b) the intermolecular force of attraction
(c) movement of particles
Advertisements
उत्तर
| Characteristics | Solids | Liquids | Gases |
| Intermolecular space | Minimum space | More than solids | Maximum space |
| Intermolecular force of attraction | Very strong | Less strong | Very weak |
| movement of particles | About their own position | In continuous motion | In any random direction |
संबंधित प्रश्न
Classify the following into solids, liquids and gases.
Oxygen, milk, common salt, wax, stone, L.P.G, carbon- dioxide, sugar, mercury, coal, blood, butter, copper, coconut oil, kerosene.
Give reasons
A red ink drop added to small amount of water in a glass turns the water red in some time.
Describe simple experiments to prove that solids have a definite volume
Describe simple experiments to prove that liquids have a definite volume
Describe simple experiments to prove that liquids have no definite shape
______ soil is suitable for growing cotton.
Particles in solids are free to move.
Strainer is a kind of sieve which is used to separate a liquid from solid.
Identify the state of matter based on the arrangement of the molecules.
Choose the odd one:
