Advertisements
Advertisements
प्रश्न
Distinguish the properties of solid, liquid, and gas. Draw a suitable diagram.
Advertisements
उत्तर
| S.No. | Solid | Liquid | Gas |
| 1. | Definite shape and volume | No definite shape, it attains the shape of the vessel | it has neither a definite shape nor a definite volume. |
| 2. | Incompressible | Compressible to a small extent | Highly compressible. |
| 3. | Little space between the particles | Greater space between the particles | Greater space between the particles. |
| 4. | Particles cannot move freely | Particles move freely | Particles are in a continuous, random motion. |
| 5. |
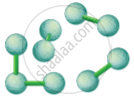
Particles in a Solid
Eg. Stone |
Particles in a Liquid
Eg. Water |
Particles in a Gas
Eg. Air |
APPEARS IN
संबंधित प्रश्न
How do solids, liquids and gases differ in shape and volume?
Give reasons
When an empty gas jar is inverted over a gas jar containing a coloured gas, the gas also spreads into the empty jar
For the following statement, say whether it describes a solid, a liquid or a gas.
Particles are quite close together
Fill the following chart showing twelve solids, twelve liquids, four gases and eight materials.
The three main states of matter are solids, liquids, and gases. Compare the three states with reference to the following characteristics of matter
(a) volume
(b) shape
(c) compressibility
(d) diffusion.
Describe simple experiments to prove that liquids have no definite shape
See the given pictures and write down the state of each object. (Solid, Liquid, Gas)
 |
 |
 |
 |
 |
| _____ | ______ | ______ | ______ | ______ |
 |
 |
 |
 |
 |
| ______ | _______ | ______ | ______ | ______ |
Complete the given analogy.
Large Inter-particle space: Gas:: ______ :solid.
What is the physical state of water at 0°C.
Solids are compressible.