Advertisements
Advertisements
प्रश्न
Complete the following table for the homologous series of alkanes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methane | CH4 | CH4 | 1 | 1 | -162 |
| Ethane | C2H6 | CH3–CH3 | 2 | 2 | -88.5 |
| Propane | C3H8 | CH3–CH2–CH3 | 3 | 3 | -42 |
| Butane | C4H10 | CH3–CH2–CH2–CH3 | ______ | ______ | 0 |
| Pentane | C5H12 | CH3–CH2–CH2–CH2–CH3 | ______ | ______ | 36 |
| Hexane | C6H14 | CH3–CH2–CH2–CH2–CH2–CH3 | ______ | ______ | 69 |
Advertisements
उत्तर
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methane | CH4 | CH4 | 1 | 1 | -162 |
| Ethane | C2H6 | CH3–CH3 | 2 | 2 | -88.5 |
| Propane | C3H8 | CH3–CH2–CH3 | 3 | 3 | -42 |
| Butane | C4H10 | CH3–CH2–CH2–CH3 | 4 | 4 | 0 |
| Pentane | C5H12 | CH3–CH2–CH2–CH2–CH3 | 5 | 5 | 36 |
| Hexane | C6H14 | CH3–CH2–CH2–CH2–CH2–CH3 | 6 | 6 | 69 |
APPEARS IN
संबंधित प्रश्न
The following vegetables are kept in a basket :
Potato, Tomato, Radish, Brinjal, Carrot, Bottle-gourd
Which two of these vegetables correctly represent the homologous structures?
(A) Carrot and Tomato
(B) Potato and Brinjal
(C) Radish and Carrot
(D) Radish and Bottle-gourd
Give the structural formula for 1, 2-dichloroethane
Fill in the following blank with suitable word:
Ethene and ethyne are examples of ..... hydrocarbons.
What is the difference between two consecutive homologues:
(1) in terms of molecular mass?
(2) in terms of number and kind of atoms per molecule?
Give the molecular formula of one homologue of the following:
C3H6
The molecular formula of an organic compound is C18H36. Name its homologous series.
Why homologous series of carbon compounds are so called? Write chemical formula of two consecutive members of a homologous series and state the part of these compounds that determines their (i) physical properties, and (ii) chemical properties.
What is a homologous series?
Haloalkanes react with alkalies to produce alcohol. Give the equation for the preparation of the second member of the homologous series of alcohol. State under what condition the reaction occurs.
Cyclohexane : Cyclic hydrocarbon : : Isobutylene : _______
Observe the structural formula and answer the following questions.
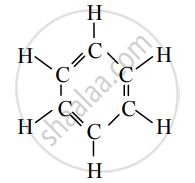
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
C3H8 belongs to the homologous series of ______.
Define Homologous series.
Consider the carbon compounds having following molecular formula:
(i) C2H2 (ii) C2H5 (iii) C3H7OH (iv) C2H6COOH (v) CH3CHO
- Identify which one of the above compounds, is a member of aldehyde series.
- Write the general formula of the series to which compound C2H2 belongs.
- Which one of the above compounds has triple bonds between carbon-carbon atoms?
- Write the molecular formula of the first member of the homologous series to which the compound C3H7OH belongs.
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H8.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
Name the third homologue of aldehydes.
