Advertisements
Advertisements
प्रश्न
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
Advertisements
उत्तर
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | 3 | 3 | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | 4 | 4 | 118 |
APPEARS IN
संबंधित प्रश्न
What is meant by homologous series of carbon compounds?
Write the molecular formula of first two members of homologous series having functional group -Cl.
The following vegetables are kept in a basket :
Potato, Tomato, Radish, Brinjal, Carrot, Bottle-gourd
Which two of these vegetables correctly represent the homologous structures?
(A) Carrot and Tomato
(B) Potato and Brinjal
(C) Radish and Carrot
(D) Radish and Bottle-gourd
By how many carbon atoms and hydrogen atoms do any two adjacent homologues differ?
Write the molecular formula of the third member of the homologous series of carbon compounds with general formula CnHO2n+1OH.
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
 .
.
Define homologous series of organic compounds. List its two characteristics. Write the name and formula of the first member of the series of alkenes.
Give three points to differentiate between saturated and unsaturated
hydrocarbons.
Copy and complete the following table, which relates to the three homologous series of hydrocarbons:
| General formula | CnH2n | CnH2n-2 | CnH2n+2 |
| IUPAC name of the homologus series | |||
| Characteristic bond type | Single bonds | ||
| IUPAC name of the first member of the series | |||
| Type of reaction with chlorine | Addition |
Complete the correlation:
Alkene : C = C :: Alkyne: _______.
Find the odd one out and give its explanation.
As one ascends in any homologous series, physical properties change gradually.
Observe the structural formula and answer the following questions.
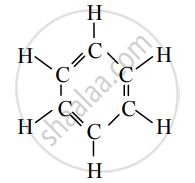
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Complete the following chart by using examples given in brackets.
(isobutylene, cyclohexane, propane, cyclohexene, cyclopentane, benzene, propyne, isobutane, propene)
| Straight chain hydrocarbons | Branched chain hydrocarbons | Cyclic hydrocarbons |
Which of the following does not belong to the same homologous series?
Successive members of a homologous series vary by how many atomic mass unit?
C3H8 belongs to the homologous series of ______.
Name the following:
Group of organic compounds where the successive members follow a regular structural pattern, successive compounds differ by a 'CH2' group.
