Advertisements
Advertisements
प्रश्न
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
Advertisements
उत्तर
Geometric isomers of [Pt(en)2ClBr]2⊕:
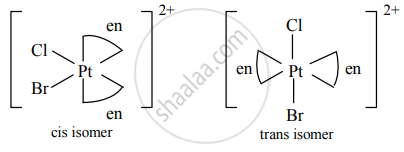
Enantiomers of [Pt(en)2ClBr]2⊕:
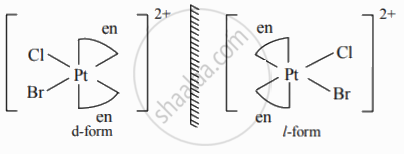
संबंधित प्रश्न
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)3]4⊕
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Which one of the following will give a pair of enantiomorphs?
Which type of isomerism is exhibited by [Pt(NH3)2Cl2]?
Explain optical isomerism in coordination compounds with an example.
What are hydrate isomers? Explain with an example.
Which of the following is NOT a pair of structural isomers?
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
How many isomers are possible for an alkane having molecular formula C5H12?
The number of geometrical isomers of [CrCl2(en)2]+ is ____________.
____________ isomers are formed when the ligand has two different donor atoms.
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
Which of the following does not show optical isomerism?
Which of the following shows maximum number of isomers?
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
Give trans isomer of [Co(NH3)4Cl2]⊕.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Explain the ionisation isomers.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Pt(NH3)4Cl2]Br2 and [Pt(NH3)4 Br2]Cl2}\]
Which one of the following complex ions has geometrical isomers?
Three organic compounds A, B and C are non cyclic functional isomers of carbonyl compounds with molecular formula C4H8O. Isomers A and C give positive Tollen’s test while compound B does not give positive Tollen’s test but gives positive iodoform test. Compounds A and B on reduction with Zn amalgam and conc. HCl give the same product.
- Write the structures of the compounds A, B and C.
- Out of the compounds A, B and C, which one will be the least reactive towards addition of HCN.
Match List-I with List-II:
| List-I (Complex) |
List-II (Type of isomerism) |
| A. [Co(NH3)5(NO2)]Cl2 | I. Solvate isomerism |
| B. [Co(NH3)5(SO4)]Br | II. Linkage isomerism |
| C. [Co(NH3)6][Cr(CN)6] | III. Ionisation isomerism |
| D. [Co(H2O)6]Cl3 | IV. Coordination isomerism |
Choose the correct answer from the options given below:
