Advertisements
Advertisements
प्रश्न
A solid mixture contains four constituents P, Q, R and S. P consists of tiny grains and it is mixed with
cement for plastering the walls. Q is a white solid which is recovered on a large scale from sea water by the
process of evaporation. R is in the form of tiny particles of a material whose corrosion is called rusting. And
S is a white solid which is used in making ordinary dry cells.
- What could P, Q, R and S be ?
- How would you separate a mixture containing P, Q, R and S ?
Advertisements
उत्तर
- P consists of tiny grains and it is mixed with cement for plastering the walls. So, P is sand. Q is a white solid, which is recovered on a large scale from sea water by the process of evaporation. It is common salt. R is in the form of tiny particles of materials, whose corrosion is called rusting. It is, iron filings. S is ammonium chloride.
- At first, let us take the mixture of P, Q, R, and S.
- We first separate R (iron filings) by using a magnet to attract them. Then separate S (ammonium chloride) by sublimation.
- Now, we shake P (sand) and Q (common salt) with water. The mixture is then filtered.
- Sand is obtained as residue.
- Now, we will evaporate the filtrate to dryness to obtain Q (common salt).
APPEARS IN
संबंधित प्रश्न
Liquids have definite volume but do not have definite shape.
Which of the following materials fall in the category of a “pure substance”?
- Ice
- Milk
- Iron
- Hydrochloric Acid
- Calcium oxide
- Mercury
- Brick
- Wood
- Air
What is the general name of the materials which contain at least two pure substances and show the properties
of their constituents ?
What are the two types of pure substances? Give one example of each type.
There is a large group of materials P which can be divided into three groups Q, R and S on the basis of their properties. The substances belonging to group Q can be solids, liquids or gases. The solids belonging to group Q are usually electrical insulators. Most of the substances of group R are solids which are good conductors of electricity. The substances belonging to group S are neither insulators like Q nor good conductors like R. The properties of S are intermediate between those of Q and R.
- What could the group of materials P be ?
- Name the substances Q. Give two examples of such substances.
- Name the substances R. Write two examples of such substances.
- Name the substances S. Give two examples of such substances.
- Out of Q, R and S, which substances are malleable and ductile ?
State whether the following statement is true or false :
Bread is an example of solid foam.
What is the difference between solutions and colloids ?
Explain what happens when a beam of light is passed through a colloidal solution.
Name the different types of solutions. Give one example of each.
Which of the following do not exhibit Tyndall effect ?
Starch solution, Sugar solution, Ink, Salt solution, Copper sulphate solution, Ammonium chloride solution, Fog, Smoke, Car exhausts.
What is a colloid ? Give two examples of colloids (or colloidal solutions)
One of the following does not show Tyndall effect. This one is :
How will you separate a mixture of common salt, sulphur powder and sand ?
Wipe the board with a duster and then tap the duster against the table. What do you see?
Justify your answer.
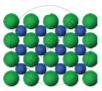
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Diffentiate between absorption and adsorption.
What type of matter glass is?
Define the term matter.
Define the term matter.
