Advertisements
Advertisements
प्रश्न
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
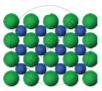
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Advertisements
उत्तर
- The liquid state is represented by Fig 1.
- In Fig. 3 the inter-particle attraction will be maximum because it is in a solid state.
- Fig. 2 cannot be contained in an open vessel, as it is in a gaseous state.
- Fig. 1 can take the shape of its container, as it is in a liquid state.
APPEARS IN
संबंधित प्रश्न
Define the term matter. What is it composed of?
Name the terms for the particles of matter which may or may not have independent existence.
A kind of matter which can sublime is
Classify the following into metals, non-metals and metalloids:
Silicon, Mercury, Diamond, Sulphur, Iodine, Germanium, Sodium, Carbon, Magnesium, Copper, Boron, Helium
Out of a colloid, solution and a suspension :
which one has the smallest particles ?
How much water should be added to 15 grams of salt to obtain 15 per cent salt solution ?
Which of the following will show Tyndall effect ? Why ?
- Salt solution
- Starch solution
- Milk
- Copper sulphate solution
What is meant by the concentration of a solution ?
Name the process which can be used to purify an impure sample of copper sulphate.
How will you separate a mixture of iron filings and powdered carbon ?
