Advertisements
Advertisements
प्रश्न
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
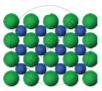 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Advertisements
उत्तर
- The liquid state is represented by Fig 1.
- In Fig. 3 the inter-particle attraction will be maximum because it is in a solid state.
- Fig. 2 cannot be contained in an open vessel, as it is in a gaseous state.
- Fig. 1 can take the shape of its container, as it is in a liquid state.
APPEARS IN
संबंधित प्रश्न
A kind of matter which can sublime is
Classify the following as elements or compounds :
Iron, Iron sulphide, Sulphur, Chalk, Washing soda, Sodium, Carbon, Urea
Which of the following substances are elements ?
Water, Salt, Mercury, Iron, Marble, Diamond, Wood, Nitrogen, Air, Graphite, Hydrogen, Oxygen, Sugar, Chlorine
Name a non-metal :
- which is lustrous
- which is required for combustion
- whose one of the allotrophic forms is a good conductor of electricity. Name the allotrope.
- other than carbon which shows allotropy
- which is known to form the largest number of compounds
Iron powder and sulphur powder were mixed together and divided into two parts A and B. When part A was heated strongly over a burner, then a substance C was formed. The part B was, however, not heated at all. When dilute hydrochloric acid was added to substance C, then gas D was evolved and when dilute hydrochloric acid was added to part B then gas E was evolved.
- What type of substance is B ?
- What type of substance is C ?
- Name the gas (i) D, and (ii) E ?
- State one characteristic property of gas D.
- Write one test to identify gas E.
The solubility of ammonium chloride in water at various temperatures is given below :
| Temperature | : | 10°C | 20°C | 40°C | 60°C | 80°C |
| Solubility | : | 24 g | 37 g | 41 g | 55 g | 66 g |
What mass of ammonium chloride would be needed to make a saturated solution of ammonium chloride in fifty grams of water at 40°C ?
How is water purified on a large scale at water works ? Explain with the help of a labelled diagram. Name the substance which is added to kill germs in the drinking water supply ?
The property which allows the metals to be hammered into their sheets is ______.
In Aerosol the dispersed phase is liquid and the dispersion medium is gas.
The phenomenon of Evaporation takes place at the ______ of a liquid.
