Advertisements
Advertisements
प्रश्न
One of the following does not show Tyndall effect. This one is :
पर्याय
soap solution
ink
sugar solution
starch solution
Advertisements
उत्तर
sugar solution
Sugar solution does not show Tyndall effect because sugar solution is a true solution and not a colloid.
APPEARS IN
संबंधित प्रश्न
Intermolecular force of attraction is maximum in ............ less in .......... and the least in .............
A kind of matter which can sublime is
Define matter. Give four examples of matter.
What are the characteristics of matter?
What is Brownian motion? Draw a diagram to show the movement of a particle (like a pollen grain during Brownian motion.
The property/properties which enable copper metal to be used for making electric wires is/are :
On the basis of composition of matter, milk is considered to be :
There is a large group of materials P which can be divided into three groups Q, R and S on the basis of their properties. The substances belonging to group Q can be solids, liquids or gases. The solids belonging to group Q are usually electrical insulators. Most of the substances of group R are solids which are good conductors of electricity. The substances belonging to group S are neither insulators like Q nor good conductors like R. The properties of S are intermediate between those of Q and R.
- What could the group of materials P be ?
- Name the substances Q. Give two examples of such substances.
- Name the substances R. Write two examples of such substances.
- Name the substances S. Give two examples of such substances.
- Out of Q, R and S, which substances are malleable and ductile ?
What is the name of the clear liquid formed when a solid dissolves in a liquid ?
Which of the following contains less solute at a given temperature and pressure ? Unsaturated solution or Saturated solution.
Name the process which can be used to recover salt from an aqueous salt solution.
Name the process which can be used to recover salt from an aqueous salt solution.
Explain how, impure copper sulphate can be purified by crystallisation.
Asmita went to buy an earthen pot. There she saw many things kept for sale.
What is the difference between a substance and an object?
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
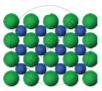 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Why wires cannot be drawn from materials such as stone or wood?
A standard sheet of paper is about ______ of atoms thick
Rusting of an article made up of iron is called
Which of the following statements are true for pure substances?
(i) Pure substances contain only one kind of particles
(ii) Pure substances may be compounds or mixtures
(iii) Pure substances have the same composition throughout
(iv) Pure substances can be exemplified by all elements other than nickel
The most reactive metal is ______.
