Advertisements
Advertisements
प्रश्न
Which of the following statements are true for pure substances?
(i) Pure substances contain only one kind of particles
(ii) Pure substances may be compounds or mixtures
(iii) Pure substances have the same composition throughout
(iv) Pure substances can be exemplified by all elements other than nickel
पर्याय
(i) and (ii)
(i) and (iii)
(iii) and (iv)
(ii) and (iii)
Advertisements
उत्तर
(i) and (iii)
Explanation:
Pure substances are made up of only one kind of particle and they have the same composition throughout. Mixtures are not pure substances. Only elements and compounds are pure substances.
APPEARS IN
संबंधित प्रश्न
Fill in the blank
All matter is made up of tiny particles called ______
When a gas jar containing air is inverted over a gas jar containing bromine vapour, the red-brown bromine vapour diffuse into air. Explain how bromine vapour diffuse into air.
What name is given to those elements which are neither good conductors of electricity like copper nor insulators like sulphur ?
What are the three groups into which all the elements can be divided ? Name two elements belonging to each group.
Give reason why graphite is used for making elecrode in a dry cell.
Classify the following into elements, compounds and mixtures :
arble, Air, Gold, Brass, Sand, Diamond, Graphite, Petroleum, Common salt, Sea-water, Chalk
In the following set of substances, one item does not belong to the set. Select this item and explain why it does not belong to the set :
Hydrogen, Oxygen, Steam, Chlorine
What is the concentration of a solution which contains 16 g of urea in 120 g of solution ?
A solution contains 5.6 mL of alcohol mixed with 75 mL of water. Calculate the concentration of this solution.
Which of the following contains less solute at a given temperature and pressure ? Unsaturated solution or Saturated solution.
Which of the following will show Tyndall effect ? Why ?
- Salt solution
- Starch solution
- Milk
- Copper sulphate solution
21.5 g of sodium chloride dissolves in 60 g of water at 25°C. Calculate the solubility of sodium chloride in water at that temperature.
What difference in the property of two miscible liquids enables their separation by fractional distillation ?
What difference in the properties of oil and water enable their separation by a separating funnel ?
Complete the given analogy.
Murukku from hot oil: ______:: Coffee powder residue from decoction: ______
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
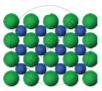 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Intermolecular space is maximum in ______.
The pressure of gases does not depend on the temperature of the gas and the volume it occupies.
Gas-solid is ______.
Evaporation is always accompanied by ______ in temperature.
