Please select a subject first
Advertisements
Advertisements
Complete and rewrite the balanced chemical equations

Concept: undefined >> undefined
Complete and rewrite the balanced chemical equations
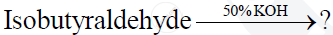
Concept: undefined >> undefined
Advertisements
Explain the cause of Lanthanoids contraction.
Concept: undefined >> undefined
How are ethylamine and ethyl methyl amine distinguished by using nitrous acid?
Concept: undefined >> undefined
What are isotonic solutions? Explain with one example.
Concept: undefined >> undefined
What are antacids?
Concept: undefined >> undefined
Derive van’t Hoff general solution equation.
Concept: undefined >> undefined
Distinguish between crystalline solids and amorsphous solids.
Concept: undefined >> undefined
Classify the following molecular solids into different types:
- HCl
- CO2
- Solid ice
- SO2
Concept: undefined >> undefined
Write any ‘four points’ of difference between fluorine and other halogens.
Concept: undefined >> undefined
Distinguish between thermoplastic polymers and thermosetting polymers.
Concept: undefined >> undefined
What is the action of the following on lanthanoids?
a. water
b. Sulphur, heat
c. nitrogen, heat
Concept: undefined >> undefined
How are the crystalline solids classified?
Concept: undefined >> undefined
Answer the following in one or two sentences.
A solid is hard, brittle and electrically nonconductor. Its melt conducts electricity. What type of solid is it?
Concept: undefined >> undefined
Answer the following in brief.
Distinguish between ionic solids and molecular solids
Concept: undefined >> undefined
Cryoscopic constant depends on ______.
Concept: undefined >> undefined
Answer in brief.
What are intensive properties? Explain why density is an intensive property.
Concept: undefined >> undefined
