Please select a subject first
Advertisements
Advertisements
Give two differences between the Disinfectant and antiseptic.
Concept: undefined >> undefined
Match the pairs.
| A group | B group |
| A. Paracetamol | a. Antibiotic |
| B. Chloramphenicol | b. Synthetic detergent |
| C. BHT | c. Soap |
| D. Sodium stearate | d. Antioxidant |
| e. Analgesic |
Concept: undefined >> undefined
Advertisements
Name two drugs which reduce body pain.
Concept: undefined >> undefined
Explain with an example of Antiseptics.
Concept: undefined >> undefined
Explain with an example Disinfectant.
Concept: undefined >> undefined
Explain with an example of Cationic detergents.
Concept: undefined >> undefined
Explain with an example of Anionic detergents.
Concept: undefined >> undefined
Explain with an example of Nonionic detergents.
Concept: undefined >> undefined
Identify the functional group in the following molecule:
Chloramphenicol
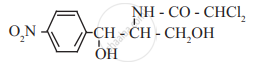
Concept: undefined >> undefined
Identify the functional group in the following molecule:
Sulphanilamide
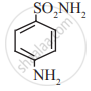
Concept: undefined >> undefined
Identify the functional group in the following molecule:
Glycerine
\[\begin{array}{cc}\ce{CH2 - OH}\\|\phantom{.........}\\\ce{CH - OH}\\|\phantom{.........}\\\ce{CH2 - OH}\end{array}\]
Concept: undefined >> undefined
What is meant by broad-spectrum antibiotic and narrow-spectrum antibiotics?
Concept: undefined >> undefined
Answer in one sentence.
Name the pain killer obtained from acetylation of salicylic acid.
Concept: undefined >> undefined
Answer in one sentence.
Name the class of drug often called as a painkiller.
Concept: undefined >> undefined
Answer in one sentence.
Draw the structure of chloroxylenol.
Concept: undefined >> undefined
Answer in one sentence.
Draw the structure of salvarsan.
Concept: undefined >> undefined
Answer in one sentence.
Write molecular formula of Butylated hydroxytoluene.
Concept: undefined >> undefined
Answer in one sentence.
What is the tincture of iodine?
Concept: undefined >> undefined
Write the molecular formula and name of:
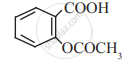
Concept: undefined >> undefined
Write two examples of the following:
- Analgesics
- Antiseptics
- Antibiotics
- Disinfectant
Concept: undefined >> undefined
