Advertisements
Advertisements
Write the balanced equation for the preparation of the following compound (as major product) starting from iron and using only one other substance:
Iron (II) chloride
Concept: undefined >> undefined
Write the balanced equation for the preparation of the following compound (as major product) starting from iron and using only one other substance:
Iron (II) sulphate
Concept: undefined >> undefined
Advertisements
The preparation of Lead sulphate from Lead carbonate is a two-step process. (Lead sulphate cannot be prepared by adding dilute Sulphuric acid to Lead carbonate.)
What is the first step that is required to prepare Lead sulphate from Lead carbonate?
Concept: undefined >> undefined
Why the alkali is added drop by drop to the salt solution?
Concept: undefined >> undefined
- Name the experiment illustrated below.
- State the colour of the water that has entered the round-bottomed flask.
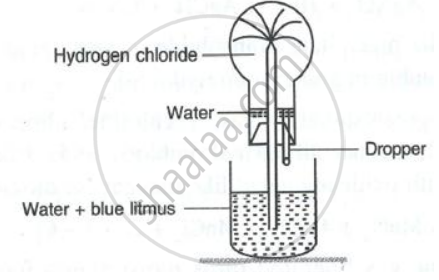
Concept: undefined >> undefined
What property of hydrogen chloride is demonstrated when it is collected by downward delivery (Upward displacement)?
Concept: undefined >> undefined
Explain with a diagram the preparation of aqueous ammonia.
Concept: undefined >> undefined
Give two balanced reactions of the type to show the following property of sulphuric acid:
Dehydrating nature
Concept: undefined >> undefined
Give a chemical test to distinguish between dilute sulphuric acid and dilute hydrochloric acid (using lead nitrate solution).
Concept: undefined >> undefined
Name the acid formed when sulphur dioxide dissolves in water.
Concept: undefined >> undefined
Name the gas released when sodium carbonate is added to a solution of sulphur dioxide.
Concept: undefined >> undefined
Name the anion present in the following compound:
Compound D, when warmed with dilute sulphuric acid, gives a gas that turns acidified dichromate solution green.
Concept: undefined >> undefined
Distinguish by a chemical test, sulphuric acid from nitric acid and hydrochloric acid.
Concept: undefined >> undefined
State your observation when lead nitrate solution and sodium chloride solution are mixed.
Concept: undefined >> undefined
State your observation when zinc chloride solution, zinc nitrate solution, and zinc sulphate solutions are added individually to barium chloride solution.
Concept: undefined >> undefined
The gas evolved when dilute sulphuric acid reacts with iron sulphide.
Concept: undefined >> undefined
An acid obtained from concentrated nitric acid on reaction with Sulphur ______.
Concept: undefined >> undefined
State the following:
The drying agent used in the laboratory preparation of HCl gas.
Concept: undefined >> undefined
Write a balanced equation for the following conversion:
Lead sulphate from lead nitrate and sulphuric acid.
Concept: undefined >> undefined
Identify the terms for the following:
A method used to collect HCl gas.
Concept: undefined >> undefined
