Advertisements
Advertisements
प्रश्न
Why the alkali is added drop by drop to the salt solution?
Advertisements
उत्तर
If an alkali is added too quickly, then it is easy to miss a precipitate which redissolves in excess alkali.
APPEARS IN
संबंधित प्रश्न
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
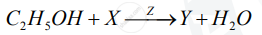
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Name two bases which are not alkalis but dissolves in strong alkalis.
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess : ZnSO4
Name: Two bases which are not alkalis but dissolve in strong alkalis.
Name the chloride of a metal which is soluble in excess of ammonium hydroxide.
Fill in the blank.
The substance/s which react with hot conc. NaOH solution and undergoes a neutralization reaction ______ [Al2O3 / Al / Al(OH)3]
When ammonium hydroxide is added to solution B, a pale blue precipitate is formed. This pale blue precipitate dissolves in excess ammonium hydroxide to give an inky blue solution. Name the cation present in solution B. What is the probable colour of solution B?
State your observation when zinc chloride solution, zinc nitrate solution, and zinc sulphate solutions are added individually to barium chloride solution.
Write balanced equations for a metal that evolves into a gas which burns with a pop sound when boiled with alkali solutions.
Write balanced equations for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
