Advertisements
Advertisements
प्रश्न
Name: Two bases which are not alkalis but dissolve in strong alkalis.
Advertisements
उत्तर
Zn(OH)2 and Pb(OH)2
APPEARS IN
संबंधित प्रश्न
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
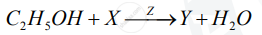
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Name
a metallic hydroxide soluble in excess of NH4OH.
Write balanced equation for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
Name the metal hydroxides which are:
Sparingly soluble
In caustic soda solution
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess : ZnSO4
What do you understand by amphoteric oxide? Give the balanced equations for the reaction with three different amphoteric oxides with a caustic alkali. Write your observation if any.
Name : A yellow monoxide that dissolves in hot and concentrated caustic alkali.
Fill in the blank.
The substance/s which react with hot conc. NaOH solution and undergoes a neutralization reaction ______ [Al2O3 / Al / Al(OH)3]
What do you see when sodium hydroxide solution is added to zinc sulphate solution, till it is in excess?
Write balanced equations for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
