Advertisements
Advertisements
Name the radiation which can be detected by thermopile.
Concept: undefined >> undefined
What is meant by the drift speed of free electrons?
Concept: undefined >> undefined
Advertisements
To which regions of the electromagnetic spectrum do the following wavelengths belong:
(a) 250 nm
(b) 1500 nm
Concept: undefined >> undefined
Arrange the following electromagnetic waves in increasing order of their frequencies (i.e. begin with the lowest frequency):
Visible light, y rays, X rays, microwaves, radio waves, infrared radiations, and ultraviolet radiation.
Concept: undefined >> undefined
Name the part of the electromagnetic spectrum which is:
Suitable for radar systems used in aircraft navigation.
Concept: undefined >> undefined
Name the part of the electromagnetic spectrum which is:
Produced by bombarding a metal target with high electrons.
Concept: undefined >> undefined
Calculate the shortest wavelength of electromagnetic radiation present in Balmer series of hydrogen spectrum.
Concept: undefined >> undefined
Write balancing condition of a Wheatstone bridge.
Concept: undefined >> undefined
Name the electromagnetic radiation whose frequency is 10 Hz.
Concept: undefined >> undefined
What is the speed of radio waves in vacuum?
Concept: undefined >> undefined
What happens when an electron collides with a positron?
Concept: undefined >> undefined
Name the electromagnetic radiation that has been used in obtaining the image below.
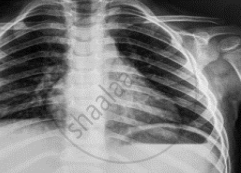
Concept: undefined >> undefined
What is the wavelength range of electromagnetic radiation used in radio broadcast?
Concept: undefined >> undefined
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.

- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
Concept: undefined >> undefined
Define equipotential surface.
Concept: undefined >> undefined
What is meant by an equipotential surface?
Concept: undefined >> undefined
An I0m long uniform metallic wire having a resistance of 20Ω IS used as a potentiometer wire. This wire is connected in series with another resistance of 480Ω
and a battery of emf 5V having negligible internal resistance. If an unknown emf e is balanced across 6m of the potentiometer wire, calculate
1) the potential gradient across the potentiometer wire
2) the value of the unknown emf e.
Concept: undefined >> undefined
Using Ampere's circuital law, obtain an expression for the magnetic flux density 'B' at a point 'X' at a perpendicular distance 'r' from a long current-carrying conductor.
(Statement of the law is not required).
Concept: undefined >> undefined
Using Ampere’s circuital law, obtain an expression for magnetic flux density ‘B’ at a point near an infinitely long and straight conductor, carrying a current I.
Concept: undefined >> undefined
A meter bridge is balanced with a known resistance (R) in the left hand gap and an unknown resistance (S) in the right hand gap. Balance point is found to be at a distance of 1 cm from the left hand side. When the battery and the galvanometer are interchanged, balance point will ______.
Concept: undefined >> undefined
