Advertisements
Advertisements
Match the terms given in Column I with the units given in Column II.
| Column I | Column II |
| (i) Λm | (a) S cm-¹ |
| (ii) ECell | (b) m-¹ |
| (iii) K | (c) S cm2 mol-¹ |
| (iv) G* | (d) V |
Concept: undefined >> undefined
Match the terms given in Column I with the items given in Column II.
| Column I | Column II |
| (i) Λm | (a) intensive property |
| (ii) ECell | (b) depends on number of ions/volume |
| (iii) K | (c) extensive property |
| (iv) ∆rGCell | (d) increases with dilution |
Concept: undefined >> undefined
Advertisements
Match the items of Column I and Column II.
| Column I | Column II |
| (i) K | (a) I × t |
| (ii) Λm | (b) `Λ_m/Λ_m^0` |
| (iii) α | (c) `K/c` |
| (iv) Q | (d) `G^∗/R` |
Concept: undefined >> undefined
Assertion: ECell should have a positive value for the cell to function.
Reason: `"E"_("cathode") < "E"_("anode")`
Concept: undefined >> undefined
Assertion: Mercury cell does not give steady potential.
Reason: In the cell reaction, ions are not involved in solution.
Concept: undefined >> undefined
The term ‘sorption’ stands for ______.
Concept: undefined >> undefined
Which one of the following is not applicable to the phenomenon of adsorption?
Concept: undefined >> undefined
Which of the following is an example of absorption?
Concept: undefined >> undefined
On the basis of data given below predict which of the following gases shows least adsorption on a definite amount of charcoal?
| Gas | CO2 | SO2 | CH1 | H2 |
| Critical temp./K | 304 | 630 | 190 | 33 |
Concept: undefined >> undefined
Which of the following process is not responsible for the presence of electric charge on the sol particles?
Concept: undefined >> undefined
Which of the following phenomenon occurs when a chalk stick is dipped in ink?
(i) Adsorption of coloured substance.
(ii) Adsorption of solvent.
(iii) Absorption and adsorption both of solvent.
(iv) Absoprtion of solvent.
Concept: undefined >> undefined
Which of the following statements are correct?
(i) A depressant prevents certain type of particle to come to the froth.
(ii) Copper matte contains \[\ce{Cu2S}\] and \[\ce{ZnS}\].
(iii) The solidified copper obtained from reverberatory furnace has blistered appearance due to evolution of \[\ce{SO2}\] during the extraction.
(iv) Zinc can be extracted by self-reduction.
Concept: undefined >> undefined
What is the role of flux in metallurgical processes?
Concept: undefined >> undefined
Identify the compound Y in the following reaction.

Concept: undefined >> undefined
In which of the following molecules carbon atom marked with asterisk (*) is asymmetric?
(a) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(b) \[\begin{array}{cc}
\ce{D}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(c) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{OH}\phantom{..}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{H}\phantom{...}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
Concept: undefined >> undefined
Which of the following structures is enantiomeric with the molecule (A) given below:
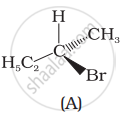
Concept: undefined >> undefined
Which of the following is an example of vic-dihalide?
Concept: undefined >> undefined
The position of \[\ce{-Br}\] in the compound in \[\ce{CH3CH = CH(Br)(CH3)2}\] can be classified as ______.
Concept: undefined >> undefined
Ethylidene chloride is a/an ______.
Concept: undefined >> undefined
Which is the correct IUPAC name for \[\begin{array}{cc}
\ce{CH3 - CH - CH2 - Br}\\
\phantom{}|\phantom{.......}\\
\phantom{}\ce{C2H5}\phantom{....}
\end{array}\]?
Concept: undefined >> undefined
