Advertisements
Advertisements
Name the device which is used as a voltage regulator. Draw the necessary circuit diagram and explain its working?
Concept: Zener Diode as a Voltage Regulator
Explain briefly the principle on which a transistor-amplifier works as an oscillator. Draw the necessary circuit diagram and explain its working ?
Concept: Junction Transistor >> Transistor as a Device
Identify the equivalent gate for the following circuit and write its truth table.
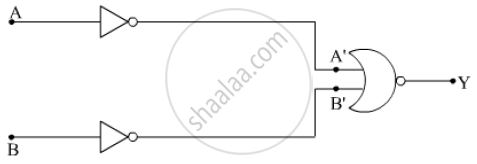
Concept: Integrated Circuits
How does a light emitting diode (LED) work? Give two advantages of LED’s over the conventional incandescent lamps.
Concept: Special Purpose P-n Junction Diodes
Draw the circuit for studying the input and output characteristics of and transistor in CE configuration. Show, how, from the output characteristics, the information about the current amplification factor (βac) can obtained.
Concept: Transistor Action
Draw a plot of the transfer characteristics (V0 versus Vi) for a base-biased transistor in CE configuration. Show for which regions in the plot, the transistor can operate as a switch ?
Concept: Transistor Action
Why is a zener diode considered as a special purpose semiconductor diode?
Concept: Semiconductor Diode
Draw the I − V characteristics of zener diode and explain briefly how reverse current suddenly increase at the breakdown voltage.
Concept: Zener Diode as a Voltage Regulator
Describe briefly with the help of a circuit diagram how a zener diode works to obtain a constant dc voltage from the unregulated dc output of a rectifier ?
Concept: Zener Diode as a Voltage Regulator
The output of an OR gate is connected to both the inputs of a NAND gate Draw the logic circuit of this combinaion of getes and write its truth table.
Concept: Digital Electronics and Logic Gates
(i) Write the truth tables of the logic gates marked P and Q in the given circuit.
(ii) Write the truth table for the circuit.
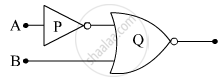
Concept: Digital Electronics and Logic Gates
Why are NOR gates considered as universal gates?
Concept: Digital Electronics and Logic Gates
Explain how a potential barrier is developed in a p-n junction diode.
Concept: Special Purpose P-n Junction Diodes
Draw the circuit arrangement for studying the V-I characteristics of a p-n junction diode in reverse bias. Plot the V-I characteristics in this case.
Concept: Special Purpose P-n Junction Diodes
Draw the V-I characteristics of an LED. State two advantages of LED lamps over convertional incandescent lamps.
Concept: Special Purpose P-n Junction Diodes
Answer the following question.
Three photodiodes D1, D2, and D3 are made of semiconductors having band gaps of 2.5 eV, 2 eV, and 3 eV respectively. Which of them will not be able to detect light of wavelength 600 nm?
Concept: Classification of Metals, Conductors and Semiconductors
Answer the following question.
Why photodiodes are required to operate in reverse bias? Explain.
Concept: Semiconductor Diode
Answer the following question.
Describe briefly the functions of the three segments of the n-p-n transistor.
Concept: Junction Transistor >> Transistor: Structure and Action
The wavelength and intensity of light emitted by a LED depend upon ______.
Concept: Special Purpose P-n Junction Diodes
The ability of a junction diode to ______ an alternating voltage is based on the fact that it allows current to pass only when it is forward biased.
Concept: Application of Junction Diode as a Rectifier
