- Carboxylic acids are carbon compounds with a –COOH group and have acidic nature.
- Types:
- Monocarboxylic acids have one –COOH group (e.g., formic acid, acetic acid)
- Dicarboxylic acids have two –COOH groups (e.g., oxalic acid) - IUPAC Naming: Replace the ‘e’ of the corresponding alkane with ‘oic acid’ (e.g., ethane → ethanoic acid); also called alkanoic acids.
Definitions [1]
Definition: Carboxylic Acid
An organic compound containing the carboxyl group (-COOH) is known as carboxylic acid. These compounds possess acidic properties.
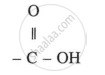
Formulae [1]
Formula: Carboxylic Acid
General formula: CnH2n+1COOH (or RCOOH)
Functional group:
Key Points
Key Points: Carboxylic Acid
Key Points: Ethanoic Acid
- Ethanoic acid (acetic acid) is a carboxylic acid; its 5–8% aqueous solution is vinegar, and it shows acidic nature by turning blue litmus red.
- Pure ethanoic acid freezes at 17 °C (290 K), so it is called glacial acetic acid.
- Being a weak acid, ethanoic acid is not completely ionised in aqueous solution.
- Ethanoic acid reacts with bases, carbonates, and bicarbonates to form a salt, water, and CO₂ (CO₂ turns lime water milky).
- It reacts with ethanol in the presence of conc. H₂SO₄ to form an ester (ethyl ethanoate) with a sweet smell (esterification).
Key Points: Physical Properties of Ethanoic Acid
- State & Smell: Colourless liquid with a strong vinegar-like smell.
- Melting Point: 17°C; forms ice-like crystals (glacial acetic acid).
- Boiling Point: 118°C.
- Nature: Hygroscopic with specific gravity 1.08.
- Solubility: Mixes well with water, alcohol, and ether.
Key Points: Chemical Properties of Ethanoic Acid
- Acetic acid is a weak acid – turns blue litmus red, reacts with metals, bases, and carbonates.
- Forms esters with alcohols in the presence of conc. H₂SO₄ (fruity smell – esterification).
- Forms acetic anhydride when heated with phosphorus pentoxide (P₂O₅).
- Reduces to ethanol using lithium aluminium hydride (LiAlH₄).
Key Points: Uses of Ethanoic Acid
- Used as a solvent for substances like phosphorus, sulphur, and cellulose.
- Acts as a laboratory reagent and is commonly used as vinegar.
- Used in dyes, perfumes, and medicines.
- Helps coagulate rubber from latex.
- Used in manufacturing cellulose acetate, amyl acetate, and acetic anhydride.
Important Questions [7]
- Reaction of an alcohol with a carboxylic acid in the presence of concentrated HA2SOA4 is termed as ______.
- Give Balanced Chemical Equations For Sodium Ethanoate to Methane.
- Name the Following: the Distinctive Reaction that Takes Place When Ethanol is Treated with Acetic Acid.
- Identify the Term Or Substance Based on the Descriptions Given Below Ice like Crystals Formed on Cooling an Organic Acid Sufficiently.
- Give a Reason For Conductivity of Dilute Hydrochloric Acid is Greater than that of Acetic Acid
- P Give the Structural Formula of Acetic Acid.
- Fill in the Blanks from the Choices Given Within Brackets: the Basicity of Acetic Acid Is-------- (3, 1, 4).
