Definitions [17]
Biomolecules are organic compounds present in living organisms as essential constituents of different cells, such as carbohydrates, proteins, fats, and amino acids.
Define peptide bond.
The bond that connects α-amino acids to each other is called a peptide bond.
Define the term Protein.
Chemically proteins are polyamides which are high molecular weight polymers of the monomer units, i.e., α-amino acids. OR It can also be defined as proteins are the biopolymers of a large number of α-amino acids and they are naturally occurring polymeric nitrogenous organic compounds containing 16% nitrogen and peptide linkages (-CO-NH-)
Define enzymes.
Enzymes are biological catalysts that speed up chemical reactions in living cells without being consumed in the process.
Define α-amino acids.
α-Amino acids are carboxylic acids having an amino (–NH2) group bonded to the α-carbon, that is, the carbon next to the carboxyl (–COOH) group.
Proteins are complex polyamides formed from amino acids. They are essential for the proper growth and maintenance of the body. They have many peptide (-CO–NH )bonds.
Chemically, proteins are polyamides, which are high molecular weight polymers of the monomer units called \[\alpha\]-amino acids.
Bifunctional organic compounds containing a carboxylic and an amino group either at the same carbon atom or at nearby carbon atoms are called amino acids.
RNA is a single-stranded nucleic acid that helps in protein synthesis and information transfer.
Define the following term.
nucleoside
The unit formed by joining the anomeric carbon of the furanose (sugar) with a nitrogen of a base is called nucleoside.
A nucleoside consists of a nitrogenous base linked to a pentose sugar without a phosphate group.
Nucleic acids are large biological macromolecules that store and transmit genetic information in living organisms.
DNA is a double-stranded nucleic acid that stores and transmits hereditary information and can replicate itself.
A nucleotide is the basic structural unit of nucleic acids, composed of a nitrogenous base, a pentose sugar, and a phosphate group.
A nitrogenous base is an organic molecule (purine or pyrimidine) that carries genetic information in nucleic acids.
Define the following as related to proteins:
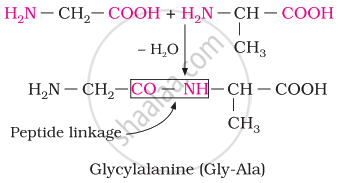
Peptide linkage
Chemically, peptide linkage is an amide formed between the –COOH group and –NH2 group. The reaction between two molecules of similar or different amino acids proceeds through the combination of the amino group of one molecule with the carboxyl group of the other. This results in the elimination of a water molecule and the formation of a peptide bond –CO–NH–. The product of the reaction is called a dipeptide because it is made up of two amino acids.
For example, when the carboxyl group of glycine combines with the amino group of alanine, we get a dipeptide, glycylalanine.
A colloidal solution of protein which works as a biological catalyst is known as an enzyme.
Key Points
- Common Composition - All living organisms are made of the same elements, like carbon, hydrogen, and oxygen, detected through elemental analysis.
- Same Elements, Different Abundance - Both living and non-living matter contain the same elements, but carbon and hydrogen are more abundant in living organisms.
- Major Elements in Human Body - Oxygen (65%), Carbon (18.5%), Nitrogen (3.3%), Hydrogen (0.5%).
- Earth's Crust vs Human Body - Silicon (27.7%) and Oxygen (46.6%) dominate Earth's crust, while Carbon dominates living matter despite being only 0.03% in the crust.
- Significance - Living organisms selectively concentrate certain elements, making their composition different from non-living matter.
- Chemical Analysis - Living tissue ground in trichloroacetic acid gives two fractions: filtrate (acid-soluble pool) containing biomolecules, and retentate (acid-insoluble fraction).
- Biomolecules - All carbon compounds derived from living tissues. Include carbohydrates, proteins, lipids, and nucleic acids.
- Amino Acids - Have –NH₂ and –COOH on the same α-carbon with a variable R group. Only 20 types occur in proteins. Categories: Acidic, Basic, Neutral.
- Lipids - Water-insoluble. Made of fatty acids + glycerol. Fatty acids: saturated (single bonds) or unsaturated (double/triple bonds).
- Nucleoside vs Nucleotide - Nucleoside = base + sugar. Nucleotide = base + sugar + phosphate group. DNA and RNA are made of nucleotides.
- Nitrogen Bases - Purines: Adenine, Guanine. Pyrimidines: Cytosine, Uracil, Thymine.
- Inorganic Components - Include Na⁺, K⁺, Ca²⁺, Mg²⁺ and compounds like NaCl, CaCO₃, PO₄³⁻, SO₄²⁻.
- Lipids are esters of fatty acids with a hydrogen-to-oxygen ratio greater than 2:1.
- They are classified into simple lipids (fats and waxes), compound lipids (phospholipids, glycolipids, lipoproteins) and sterols (derived lipids).
- Simple lipids are esters of fatty acids with various alcohols, while compound lipids typically contain 1 glycerol, 2 fatty acids and either 1 phosphate group (phospholipid) or 1 simple sugar (glycolipid).
- Glycolipids, also called cerebrosides, are abundant in the myelin sheath of nerve cells.
- In plants, sterols occur as phytosterols; the yam plant (Dioscorea) yields the sterol diosgenin, used to manufacture birth‑control pills.
- Primary Metabolites - Compounds required for basic life processes like photosynthesis, respiration, and protein metabolism. Examples: amino acids, sugars. Found in all animal tissues.
- Secondary Metabolites - Compounds produced mainly by plants, fungi, and microbes with no direct role in growth and development. Examples: alkaloids, rubber, essential oils, antibiotics, pigments, gums, spices.
- Examples by Category - Pigments: Carotenoids, Anthocyanins; Alkaloids: Morphine, Codeine; Terpenoids: Monoterpenes, Diterpenes; Toxins: Abrin, Ricin; Lectins: Concanavalin A; Drugs: Vinblastin, Curcumin; Polymeric substances: Rubber, Gums, Cellulose.
- Importance - Many secondary metabolites are useful to human welfare (e.g., rubber, drugs, spices, pigments), and some have ecological importance.
- Unknown Roles - The role of many secondary metabolites in host organisms is not fully understood.
- Biomacromolecules in the acid-insoluble fraction include proteins, nucleic acids, polysaccharides, and lipids.
- Most biomacromolecules (except lipids) have high molecular weight (≥10,000 Da) and are generally polymeric in nature.
- Micromolecules have a molecular weight of less than 1000 Da, while macromolecules have a molecular weight of thousands or more.
- Lipids are placed in the acid-insoluble fraction even though they have low molecular weight because they are part of cell membranes.
- The acid-insoluble fraction mainly represents the cytoplasmic and organelle composition of cells.
- Water is the most abundant component of cells (70–90%), followed by proteins, nucleic acids, carbohydrates, lipids, and ions.
- Together, the acid-soluble and acid-insoluble fractions represent the complete chemical composition of living tissues.
- Proteins are polymers of amino acids (polypeptides) in which amino acids are linked by peptide bonds.
- There are 20 types of amino acids, so proteins are heteropolymers (not homopolymers).
- Amino acids are of two types: essential (must be obtained from diet) and non-essential (can be synthesised in the body).
- Proteins are high molecular weight biomolecules (polyamides) made of α-amino acids with a general structure R-CH(NH₂)-COOH.
- Proteins perform various functions such as enzymatic activity, transport, hormonal regulation, immunity, and sensory reception.
- Proteins are of two main types: fibrous proteins (insoluble, structural, e.g., keratin) and globular proteins (soluble, functional, e.g., enzymes, insulin).
- Collagen is the most abundant protein in animals, while RuBisCO is the most abundant enzyme in the biosphere.
- Starch — A polymer of α-D-glucopyranose. It has two components: amylose (α-1,4-glycosidic linkage) and amylopectin (both α-1,4 and α-1,6-glycosidic linkages).
- Cellulose — A polymer of β-glucopyranose units linked by β-1,4-glycosidic bonds.
- Glycogen — A polymer of glucose units.
- Linkage Comparison —
Polysaccharide Monomer Linkage Amylose (Starch) α-D-glucopyranose α-1,4 Amylopectin (Starch) α-D-glucopyranose α-1,4 and α-1,6 Cellulose β-glucopyranose β-1,4 Glycogen Glucose — - Key Distinction — Starch and Cellulose are both glucose polymers but differ in linkage type: Starch has α-glycosidic bonds (digestible by humans), while Cellulose has β-glycosidic bonds (not digestible by humans).
- Nucleic acids are biomacromolecules present in the acid-insoluble fraction and are responsible for the storage and transmission of genetic information (DNA and RNA).
- They are polynucleotides, formed by repeated units called nucleotides.
- Each nucleotide consists of three components: a nitrogenous base, a pentose sugar, and a phosphate group.
- Nitrogenous bases are of two types: purines (adenine, guanine) and pyrimidines (cytosine, thymine, uracil).
- The sugar present is either ribose (in RNA) or 2′-deoxyribose (in DNA).
- DNA is double-stranded and contains bases A, T, G, and C, while RNA is single-stranded and contains A, U, G, and C.
- DNA stores genetic information, while RNA plays a key role in protein synthesis and the expression of genetic information.
| Level | Description | Stabilising Forces |
|---|---|---|
| Primary | Linear sequence of amino acids linked by peptide bonds | Peptide bonds |
| Secondary | α-helix (right-handed coil, –NH of one AA H-bonded to C=O of 4th residue) OR β-pleated sheet (when R group is small) | Hydrogen bonds |
| Tertiary | Further folding of secondary structure; 3D shape | Hydrophobic interactions, H-bonds, disulphide bonds, van der Waals, ionic interactions |
| Quaternary | Two or more polypeptide chains arranged spatially | All forces present in tertiary structure |
- Enzymes are biological catalysts, mostly proteins, that increase the rate of biochemical reactions without being consumed.
- Some enzymes are ribozymes, which are RNA molecules that act like enzymes.
- Enzymes have primary, secondary, and tertiary structures, and their 3D structure determines their specificity and function.
- Each enzyme has a specific active site where the substrate binds to form an enzyme–substrate complex.
- Enzymes are highly specific and lower the activation energy of reactions.
- Enzyme activity is affected by temperature and pH; most enzymes are denatured at high temperatures, while thermophilic enzymes remain stable at 80–90°C.
- Examples of enzymes include amylase (starch → glucose), pepsin (proteins → amino acids), lactase (lactose → glucose + galactose), and maltase (maltose → glucose).
Mechanism of Enzyme Action (Lock and Key model):
- Enzyme (E) binds to substrate (S) → ES complex (E + S → ES)
- Product formation: ES → EP
- Product released: EP → E + P (enzyme regenerated)
- Enzymes work best at 298 K to 313 K (25°C to 40°C) — optimum temperature
- Activity decreases with temperature increase or decrease beyond optimum range; stops at ~273 K
- Chemical compounds undergo two types of changes: physical changes (no bond breaking, e.g., melting) and chemical reactions (bonds break and new bonds form).
- The rate of a reaction is defined as the amount of product formed per unit time, and it increases with temperature.
- A general rule is that the rate of a reaction doubles or halves with every 10°C change in temperature.
- Catalysed reactions occur much faster than uncatalysed ones, and enzymes greatly increase reaction rates.
- Enzyme-catalysed reactions can speed up processes by millions of times (e.g., carbonic anhydrase reaction).
- A metabolic pathway is a series of enzyme-catalysed reactions where each step is controlled by specific enzymes.
- The same metabolic pathway can produce different end products under different conditions (e.g., pyruvate, lactic acid, or ethanol).
- Substrate & Product - Chemical converted by enzyme = substrate (S); result = product (P). Enzymes are proteins with an active site.
- ES Complex - Substrate binds to the active site, forming a temporary Enzyme-Substrate (ES) complex. E + S → ES → EP → E + P
- Transition State - The substrate must pass through an unstable transition state structure to become the product.
- Activation Energy - Energy difference between S and transition state = activation energy. Enzymes lower this barrier, making reactions faster.
- Exothermic vs Endothermic - P at lower energy than S = exothermic (no heating needed). P at higher energy = endothermic (needs energy).
- Key Role - Enzymes reduce activation energy → increase reaction rate, regardless of reaction type.
- ES Complex Formation - Substrate (S) binds to the active site of the enzyme (E) to form a highly reactive but short-lived Enzyme-Substrate (ES) complex.
- Induced Fit - Binding of substrate induces the enzyme to alter its shape, fitting more tightly around the substrate.
- Bond Breaking - The active site, now close to the substrate, breaks the chemical bonds of the substrate, forming an Enzyme-Product (EP) complex.
- Product Release - The enzyme releases the product (P) and returns to its original free form, ready to bind another substrate molecule.
- Catalytic Cycle - E + S → ES → EP → E + P. The enzyme is unchanged at the end and repeats the cycle again and again.
- Increasing substrate concentration raises enzyme activity only up to a maximum, after which the rate levels off because all active sites become saturated.
- Increasing enzyme concentration generally increases the reaction rate, as more active sites are available for substrate binding.
- Enzyme activity is highest at an optimum temperature; high temperatures denature enzymes (destroy higher‑level structure), while low temperatures reduce their activity.
- Each enzyme has its own optimum pH range; outside this range, activity falls sharply and the enzyme may not function.
- Co‑enzymes, activators and inhibitors also affect enzyme activity: activators (often inorganic ions) enhance activity, inhibitors decrease it, and many enzymes function as a combination of apoenzyme plus co‑enzyme.
- 6 Classes of Enzymes - Enzymes are classified into 6 classes based on the type of reaction they catalyse. Each class has 4-13 subclasses, and enzymes are named by a four-digit number.
- Oxidoreductases/Dehydrogenases - Catalyse oxidation-reduction reactions between two substrates (S and S').
- Transferases, Hydrolases & Lyases - Transferases: transfer a group (G) between two substrates. Hydrolases: catalyse the hydrolysis of ester, ether, peptide, and glycosidic bonds. Lyases: remove groups by mechanisms other than hydrolysis, forming double bonds.
- Isomerases & Ligases - Isomerases: catalyse inter-conversion of isomers (optical, geometric, positional). Ligases: catalyse the linking/joining of two compounds via bonds.
- Meaning - Non-protein constituents that bind to the apoenzyme to make it catalytically active. Apoenzyme + Cofactor = active enzyme.
- Prosthetic Groups - Tightly bound organic compounds. Example: Haem in peroxidase and catalase.
- Co-enzymes - Temporarily associated organic compounds; many contain vitamins. Example: NAD and NADP contain vitamin Niacin.
- Metal Ions - Form coordination bonds at the active site. Example: Zinc in Carboxypeptidase.
- Key Distinction - Prosthetic = permanently bound | Co-enzymes = temporarily bound | Metal ions = coordination bonds.
Concepts [16]
- Concept of Biomolecules
- Analysis of Chemical Composition
- Biomolecules in the Cell > Lipids
- Primary and Secondary Metabolites
- Biomacromolecules
- Biomolecules in the Cell > Proteins
- Polysaccharides
- Biomolecules in the Cell > Nucleic Acids
- Structure of Proteins
- Biomolecules in the Cell > Enzymes
- Enzymes - Chemical Reactions
- Enzyme- High Rates Chemical Conversion
- Nature of Enzyme Action
- Factors Affecting Enzyme Activity
- Classification and Nomenclature of Enzymes
- Enzymes - Co-factors
