Definitions [22]
In term of electron transfer, define
Reduction
Reduction is defined as the phenomenon in which an atom gains an electron to form a negatively charged ion called an anion.
During the formation of ionic bond one atom undergoes oxidation while another atom undergoes reduction.
A chemical bond may be defined as the force of attraction between any two atoms in a molecule to maintain stability.
or
The phenomenon of union of two or more atoms by redistribution of electrons, so that each atom involved in bonding acquires stable configuration to gain stability or to achieve a state of lower energy is called chemical bonding.
Define a chemical bond.
A chemical bond may be defined as the force of attraction between any two atoms, in a molecule, to maintain stability.
In term of electron transfer, define Oxidation
Oxidation is the loss of electrons during a reaction by a molecule, atom or ion. In terms of electron transfer, oxidation is defined as the phenomenon in which an atom loses an electron to form a positively charged cation.
During the formation of ionic bond one atom undergoes oxidation while another atom undergoes reduction.
Define octet rule.
Octet rule: Atoms of elements combine with each other in order to complete their respective octets so as to acquire the stable gas configuration.
The octet rule or the electronic theory of chemical bonding was developed by Kossel and Lewis. According to this rule, atoms can combine either by transfer of valence electrons from one atom to another or by sharing their valence electrons in order to attain the nearest noble gas configuration by having an octet in their valence shell.

The octet rule successfully explained the formation of chemical bonds depending upon the nature of the element.
A metallic element, whose one atom readily loses electron(s) to form a positively charged ion, is an electropositive element.
The chemical compounds formed as a result of the transfer of electrons from one atom of an element to one atom of another element are called ionic (or electrovalent) compounds.
A metallic atom, which loses electron(s), becomes a positively charged ion and is known as a cation.
An ion is a charged particle which is formed due to the gain or the loss of one or more electrons by an atom.
A non-metallic atom, which gains electron(s), becomes a negatively charged ion and is known as an anion.
The number of electrons that an atom of an element loses or gains to form a electrovalent bond is called its electrovalency.
The cation and the anion being oppositely charged attract each other and form a chemical bond. Since this chemical bond formation is due to the electrostatic force of attraction between a cation and an anion, it is called an electrovalent (or an ionic) bond.
A non-metallic element, whose atom readily accepts electron(s) to form a negatively charged ion, is an electronegative element.
Define bond order.
The number of bonds formed between the two bonded atoms in a molecule is called the bond order.
Bond order = `("N"_"b" - "N"_"a")/2`
Define bond energy.
The bond enthalpy is defined as the minimum amount of energy required to break one mole of a particular bond in molecules in their gaseous state. The unit of bond enthalpy is kJ mol-1.
Define the bond length.
Bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule.
Bond lengths are expressed in terms of Angstrom (10–10 m) or picometer
(10–12 m) and are measured by spectroscopic X-ray diffractions and electron-diffraction techniques.
In an ionic compound, the bond length is the sum of the ionic radii of the constituting atoms (d = r+ + r–). In a covalent compound, it is the sum of their covalent radii (d = rA+ rB).
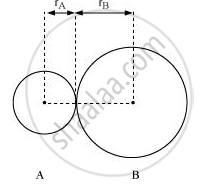
Bond-length: It is the equilibrium distance between the nuclei of two bonded atoms in a molecule. Bond-lengths are measured by spectroscopic methods
Define electronegativity.
Electronegativity is the ability of an atom in a chemical compound to attract a bond pair of electrons towards itself.
Electronegativity of any given element is not constant. It varies according to the element to which it is bound. It is not a measurable quantity. It is only a relative number
Electronegativity is the tendency of an atom to attract shared pair of electrons. It is the property of bonded atom.
The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is called its electronegativity.
Define a covalent bond.
The chemical bond formed between two combining atoms by mutual sharing of one or more pairs of electrons is called a covalent bond.
Define a coordinate bond.
The bond formed between two atoms by sharing a pair of electrons, provided entirely by one of the combining atoms but shared by both, is called a coordinate bond.
The chemical bond that is formed between two combining atoms by mutual sharing of one or more pairs of electrons is called a covalent (or a molecular) bond, and the compound formed due to this bond is called a covalent compound.
The molecule formed due to the sharing of electrons (covalent bond) is called a covalent molecule.
Formulae [4]
\[\mathrm{Bond~Order}=\frac{N_b-N_a}{2}\]
where Nb = number of electrons in bonding MOs, Na = number of electrons in antibonding MOs.
-
Bond order > 0 → molecule is stable
-
Bond order = 0 or negative → molecule is unstable (does not exist)
\[\mu=\sqrt{n(n+2)}\text{BM (Bohr Magneton)}\]
where n = number of unpaired electrons. If any unpaired electron is present → paramagnetic; if none → diamagnetic.
\[\mathrm{Bond~Order}=\frac{N_b-N_a}{2}\]
where Nb = number of electrons in bonding MOs, Na = number of electrons in antibonding MOs.
-
Bond order > 0 → molecule is stable
-
Bond order = 0 or negative → molecule is unstable (does not exist)
\[\mu=\sqrt{n(n+2)}\text{BM (Bohr Magneton)}\]
where n = number of unpaired electrons. If any unpaired electron is present → paramagnetic; if none → diamagnetic.
Theorems and Laws [1]
- Proposed by Heitler and London (1927), further developed by Pauling and Slater.
- A covalent bond is formed when half-filled valence atomic orbitals of similar energies overlap, each containing one unpaired electron.
- Greater the overlap → stronger the bond.
Types of Orbital Overlap:
| Type | Description | Bond Formed |
|---|---|---|
| Axial (Head-on) overlap | Orbitals overlap along the internuclear axis | Sigma (σ) bond |
| Sidewise (Lateral) overlap | Orbitals overlap parallel to each other, perpendicular to the internuclear axis | Pi (π) bond |
Hybridisation & Shapes:
| Hybridisation | Shape | Coordination No. |
|---|---|---|
| sp³ | Tetrahedral | 4 |
| dsp² | Square planar | 4 |
| sp³d | Trigonal bipyramidal | 5 |
| d²sp³ | Octahedral (inner) | 6 |
| sp³d² | Octahedral (outer) | 6 |
Limitations of VBT:
- Involves a number of assumptions.
- Does not give a quantitative interpretation of magnetic data.
- Does not explain the colour exhibited by coordination compounds.
- Does not give a quantitative interpretation of the thermodynamic or kinetic stabilities of coordination compounds.
- Does not make exact predictions regarding the tetrahedral and square planar structures of 4-coordinate complexes.
- Does not distinguish between weak and strong ligands.
Key Points
In 1916, Kossel and Lewis independently proposed a theory of chemical combination.
- Atoms of different elements take part in chemical combination to complete their octet (8 electrons) or duplet (2 electrons) in the outermost shell.
- All valence shell (outer-shell) electrons of atoms are represented in Lewis structures using dots surrounding the element symbol.
- Lewis structures show only valence electrons of each atom — inner shell electrons are not shown.
Carbon, nitrogen, oxygen, and fluorine always obey the octet rule in their stable compounds. However:
- Second-row elements like B and Be often have fewer than 8 electrons (incomplete octet).
- Third-row elements can exceed 8 electrons (expanded octet) using d-orbitals.
- Octet Rule: Atoms tend to gain, lose, or share electrons so that their outermost shell attains 8 electrons (like a noble gas configuration).
- Duplet Rule: Hydrogen and lithium attain stability with only 2 electrons (like helium).
- The octet rule explains why most main-group elements form bonds in fixed ratios.
- A covalent bond is formed by the mutual sharing of electron pairs between two atoms.
- A single bond shares 1 pair (2 electrons); a double bond shares 2 pairs; a triple bond shares 3 pairs.
- Co-ordinate (Dative) Bond: A special covalent bond in which both electrons are donated by one atom (the donor) to another (the acceptor). Shown as X:→Y.
- Bond angle: The angle between orbitals containing bonding pairs around the central atom. E.g., bond angle of water = 104.5°.
- Bond enthalpy (Bond energy): Energy required to break one mole of a bond in the gaseous state. E.g., H–H bond enthalpy = 435.8 kJ mol⁻¹.
Lewis structures use dots (lone pairs) and dashes (bonds) to represent all valence electrons in a molecule. Key examples:
| Molecule/Ion | Description |
|---|---|
| H₂ | H:H — single bond, no lone pairs |
| O₂ | :O::O: — double bond with lone pairs on each O |
| O₃ | One double and one single bond; resonance hybrid |
| NF₃ | N with one lone pair; 3 N−F single bonds |
| CO₃²⁻ | Resonance among 3 equivalent structures; charge = −2 |
| HNO₃ | N forms double bond with one O; OH and another O attached |
Steps to draw Lewis structure:
- Count total valence electrons (add electrons for negative charge; subtract for positive)
- Arrange atoms — least electronegative atom is usually in the centre
- Connect atoms with single bonds
- Complete octets on outer atoms first, then on central atom
- If central atom has deficit, form multiple bonds
Formal charge is a bookkeeping tool — it is the hypothetical charge on an atom in a Lewis structure assuming electrons in bonds are equally shared. It helps identify the most stable (lowest energy) Lewis structure.
\[F.C.=V.E.-N.E.-\frac{B.E.}{2}\]
where:
- V.E. = Total number of valence electrons of the atom in a free state
- N.E. = Total number of non-bonding (lone pair) electrons on that atom
- B.E. = Total number of bonding (shared) electrons around that atom
Key rules:
- The sum of formal charges in a neutral molecule = 0
- The sum of formal charges in an ion = charge of that ion
- The most stable Lewis structure has formal charges as close to zero as possible
- Negative formal charge should be on the more electronegative atom
Example — CO₃²⁻ and Ozone (O₃): Both have multiple valid Lewis structures (resonance), and formal charges help identify the preferred one.
The octet rule is a useful guideline but not universal. Three important exceptions:
| Exception | Description | Examples |
|---|---|---|
| Incomplete octet | Central atom has fewer than 8 electrons | BF₃, BeCl₂, LiCl |
| Expanded octet | Central atom has more than 8 electrons (uses d-orbitals; only elements from Period 3 and beyond) | SF₆, PCl₅, H₂SO₄, ClF₃ |
| Odd-electron molecules | Molecule has an odd number of electrons; one electron is unpaired | NO, NO₂ |
An ionic bond is formed by the complete transfer of one or more electrons from an electropositive atom to an electronegative atom, resulting in oppositely charged ions that attract each other.
Key conditions for ionic bond formation:
-
One atom must have low ionisation enthalpy (easily loses electron) — typically a metal
-
The other must have high electron affinity (easily gains electron) — typically a non-metal
-
Large difference in electronegativity between the two atoms
Example: Na + Cl → Na⁺ + Cl⁻ → NaCl
-
Sodium (2,8,1) loses 1 electron → Na⁺ (2,8)
-
Chlorine (2,8,7) gains 1 electron → Cl⁻ (2,8,8)
Ionic solids are crystalline structures containing cations and anions held together by strong electrostatic ionic bonds.
- Distance between nuclei of two bonded atoms at equilibrium.
- Inversely proportional to bond order.
- Trend: Triple < Double < Single (shortest → longest).
- Depends on the size of atoms (larger atoms → longer bond length).
- Measured in picometres (pm) or Ångström (Å) (1 Å = 100 pm).
- Greater bond length → weaker bond.
- Angle between two adjacent bonds at the central atom.
- Determined by the shape of the molecule (VSEPR theory).
- Repulsion order: LP–LP > LP–BP > BP–BP.
- More lone pairs → smaller bond angle.
- Examples: CH₄ (109.5°), NH₃ (107°), H₂O (104.5°).
- Affected by electronegativity and hybridisation.
- Energy required to break one mole of bonds in the gaseous state.
- Directly proportional to bond strength.
- Increases with an increase in bond order.
- Triple bond > Double bond > Single bond (enthalpy trend).
- Depends on bond length (shorter bond → higher enthalpy).
- Measured in kJ mol⁻¹.
- Number of bonds between two atoms.
-
Formula (MO theory): \[\frac{N_b-N_a}{2}\]
- Higher bond order → stronger and shorter bond.
- Determines the stability of a molecule.
- Examples: F₂ = 1, O₂ = 2, N₂ = 3.
- CO and NO⁺ have a bond order = 3.
- Resonance occurs when a single Lewis structure cannot accurately describe a molecule.
- Multiple structures (canonical structures) differing only in the arrangement of electrons (not atoms) are drawn.
- The actual molecule is a resonance hybrid — a weighted average of all canonical structures.
- Resonance energy = Actual bond energy − Energy of most stable resonating structure.
Conditions for Writing Resonance Structures:
- Same atomic positions in all structures.
- Same number of unpaired electrons.
- Nearly the same energy.
- Negative charge on the more electronegative atom; positive charge on the electropositive atom.
- Like charges should not reside on adjacent atoms.
Examples:
| Molecule/Ion | No. of Resonance Structures | Feature |
|---|---|---|
| O₃ | 2 | Each O–O bond is intermediate (1.5 order) |
| CO₃²⁻ | 3 | All C–O bonds are equivalent |
| NO₂⁻ | 2 | Both N–O bonds equivalent |
| Benzene (C₆H₆) | 2 | All C–C bonds equivalent |
| SO₄²⁻ | Multiple | Equivalent S–O bonds |
| Type | Description | Examples |
|---|---|---|
| Non-polar Covalent | Same atoms; electron pair lies exactly midway | H₂, O₂, Cl₂, N₂ |
| Polar Covalent | Different atoms; electron pair shifts toward the more electronegative atom | HCl, H₂O, NH₃ |
- Proposed by Sidgwick and Powell (1940) and further developed by Nyholm and Gillespie.
- The geometry of a molecule depends on the total number of valence shell electron pairs (bond pairs + lone pairs) around the central atom.
- Electron pairs repel each other and arrange themselves as far apart as possible to minimise repulsion.
- Repulsion order:
lp–lp > lp–bp > bp–bp, and lone pairs occupy more space than bond pairs. - Presence of lone pairs reduces bond angle; if no lone pairs → molecular geometry = electron pair geometry.
VSEPR Geometry Table:
| Electron Pairs | Lone Pairs | Bonding Pairs | Electron Pair Geometry | Molecular Geometry | Examples |
|---|---|---|---|---|---|
| 2 | 0 | 2 | Linear | Linear | BeBr₂, CO₂ |
| 3 | 0 | 3 | Trigonal planar | Trigonal planar | BF₃, BCl₃, BH₃ |
| 4 | 0 | 4 | Tetrahedral | Tetrahedral | CH₄, NH₄⁺, SiCl₄ |
| 5 | 0 | 5 | Trigonal bipyramidal | Trigonal bipyramidal | PCl₅, SbF₅, AsF₅ |
| 6 | 0 | 6 | Octahedral | Octahedral | SF₆, TeF₆, SeF₆ |
| 3 | 1 | 2 | Trigonal planar | Bent | SO₂, O₃ |
| 4 | 1 | 3 | Tetrahedral | Trigonal pyramidal | NH₃, PCl₃ |
| 4 | 2 | 2 | Tetrahedral | Bent | H₂O, OF₂, H₂S, SCl₂ |
| 5 | 1 | 4 | Trigonal bipyramidal | See-saw | SF₄ |
| 5 | 2 | 3 | Trigonal bipyramidal | T-shaped | ClF₃, BrF₃, ICl₃ |
| 6 | 1 | 5 | Octahedral | Square pyramidal | BrF₅, IF₅ |
| 6 | 2 | 4 | Octahedral | Square planar | XeF₄ |
Hybridisation is the process of mixing orbitals of nearly similar energy from the same atom to form a new set of equivalent orbitals of exactly equal energy called hybrid orbitals.
\[H=\frac{1}{2}[V+Y-C+A]\]
where V = valence electrons of central metal atom, Y = number of monovalent atoms surrounding central atom, C = total positive charge, A = total negative charge on the molecule.
Characteristics of Hybridisation:
- Number of hybridised orbitals = number of orbitals that participated in hybridisation.
- Hybridised orbitals are always equivalent in energy and shape.
- Hybrid orbitals are more effective in forming stable bonds than pure atomic orbitals.
- Hybrid orbitals are directed in space in some preferred directions → determines geometry of the molecule.
Hybridisation is the process of mixing orbitals of nearly similar energy from the same atom to form a new set of equivalent orbitals of exactly equal energy called hybrid orbitals.
\[H=\frac{1}{2}[V+Y-C+A]\]
where V = valence electrons of central metal atom, Y = number of monovalent atoms surrounding central atom, C = total positive charge, A = total negative charge on the molecule.
Characteristics of Hybridisation:
- Number of hybridised orbitals = number of orbitals that participated in hybridisation.
- Hybridised orbitals are always equivalent in energy and shape.
- Hybrid orbitals are more effective in forming stable bonds than pure atomic orbitals.
- Hybrid orbitals are directed in space in some preferred directions → determines geometry of the molecule.
| Hybridisation | Orbitals Mixed | Shape | Bond Angle | Example |
|---|---|---|---|---|
| sp | 1s + 1p | Linear | 180° | BeCl₂, C₂H₂ |
| sp² | 1s + 2p | Trigonal planar | 120° | BF₃, BCl₃ |
| sp³ | 1s + 3p | Tetrahedral | 109°28′ | CH₄, NH₃, H₂O |
| sp³d | 1s + 3p + 1d | Trigonal bipyramidal | 90°, 120° | PCl₅, SF₄ |
| sp³d² | 1s + 3p + 2d | Octahedral | 90° | SF₆, XeF₄ |
Molecular orbitals (MOs) are formed by the linear combination of atomic orbitals (LCAO).
Two types of MOs form:
- Bonding MOs — lower energy than the original atomic orbitals; electrons here stabilise the molecule (σ, π)
- Antibonding MOs — higher energy; electrons here destabilise the molecule (σ*, π*)
Energy Order of MOs for Diatomic Molecules:
For O₂, F₂ (electrons > 14):
For B₂, C₂, N₂ (electrons ≤ 14):
Electronic Configurations and Bond Properties of Diatomic Molecules:
| Molecule | Electronic Configuration | Bond Order | Magnetic Nature |
|---|---|---|---|
| H₂ | (σ1s)² | 1 | Diamagnetic |
| Li₂ | (σ1s)²(σ1s)²(σ2s)² | 1 | Diamagnetic |
| N₂ | (σ1s)²(σ1s)²(σ2s)²(σ2s)²(π2px)²(π2py)²(σ2pz)² | 3 | Diamagnetic |
| O₂ | (σ1s)²(σ1s)²(σ2s)²(σ2s)²(σ2pz)²(π2px)²(π2py)²(π2px)¹(π2py)¹ | 2 | Paramagnetic |
| F₂ | (σ1s)²(σ1s)²(σ2s)²(σ2s)²(σ2pz)²(π2px)²(π2py)²(π2px)²(π2py)² | 1 | Diamagnetic |
Molecular orbitals (MOs) are formed by the linear combination of atomic orbitals (LCAO).
Two types of MOs form:
- Bonding MOs — lower energy than the original atomic orbitals; electrons here stabilise the molecule (σ, π)
- Antibonding MOs — higher energy; electrons here destabilise the molecule (σ*, π*)
Energy Order of MOs for Diatomic Molecules:
For O₂, F₂ (electrons > 14):
For B₂, C₂, N₂ (electrons ≤ 14):
Electronic Configurations and Bond Properties of Diatomic Molecules:
| Molecule | Electronic Configuration | Bond Order | Magnetic Nature |
|---|---|---|---|
| H₂ | (σ1s)² | 1 | Diamagnetic |
| Li₂ | (σ1s)²(σ1s)²(σ2s)² | 1 | Diamagnetic |
| N₂ | (σ1s)²(σ1s)²(σ2s)²(σ2s)²(π2px)²(π2py)²(σ2pz)² | 3 | Diamagnetic |
| O₂ | (σ1s)²(σ1s)²(σ2s)²(σ2s)²(σ2pz)²(π2px)²(π2py)²(π2px)¹(π2py)¹ | 2 | Paramagnetic |
| F₂ | (σ1s)²(σ1s)²(σ2s)²(σ2s)²(σ2pz)²(π2px)²(π2py)²(π2px)²(π2py)² | 1 | Diamagnetic |
The electrostatic force of attraction between a hydrogen atom (covalently bonded to a highly electronegative atom) and an electronegative atom (F, O, or N) of the same or different molecule.
- Hydrogen bonding is possible when H is attached to N, O, or F (highly electronegative and small atoms).
- A hydrogen bond is weaker than a covalent bond but stronger than van der Waals forces.
The electrostatic force of attraction between a hydrogen atom (covalently bonded to a highly electronegative atom) and an electronegative atom (F, O, or N) of the same or different molecule.
- Hydrogen bonding is possible when H is attached to N, O, or F (highly electronegative and small atoms).
- A hydrogen bond is weaker than a covalent bond but stronger than van der Waals forces.
- H is bonded to a highly electronegative atom (F, O, N) → H becomes highly positive (δ+).
- This δ+ H is attracted to the lone pair of an electronegative atom of another molecule.
| Feature | Intermolecular H-Bond | Intramolecular H-Bond |
|---|---|---|
| Location | Between two different molecules of the same or different substances | Within the same molecule (between H and an electronegative atom of the same molecule) |
| Example | HF molecules, H₂O molecules, alcohol molecules | ortho-nitrophenol, ortho-chlorophenol |
| Effect | Raises boiling point significantly | Lowers the boiling point compared to intermolecular |
- Electronegativity is the ability of an atom to pull shared electrons; it is highest for fluorine (4.0).
- It increases across a period (left to right) and decreases down a group (top to bottom).
- Non-metals have high electronegativity (gain electrons), while metals have low electronegativity (lose electrons).
- Greater electronegativity difference increases ionic character.
- Carbon forms covalent bonds by sharing electrons to achieve a noble gas configuration.
- Covalent bonds can be single, double, or triple, as seen in molecules like H₂, O₂, and N₂.
- Covalent compounds have low melting and boiling points and are poor conductors of electricity.
- Carbon has allotropes such as diamond, graphite, and fullerene (C₆₀), each with different physical properties.
Concepts [44]
- Concept of Chemical Bonding
- Kossel and Lewis Approach to Chemical Bonding
- Kossel-lewis Approach to Chemical Bonding - Octet Rule
- Kossel-lewis Approach to Chemical Bonding - Covalent Bond
- Lewis Structures (Lewis Representation of Simple Molecules)
- Formal Charge
- Limitations of the Octet Rule
- Ionic or Electrovalent Bond
- Bond Parameters
- Bond Length
- Bond Angle
- Bond Enthalpy
- Bond Order
- Resonance Structures
- Polarity of Bonds
- Valence Shell Electron Pair Repulsion (VSEPR) Theory
- Valence Bond Theory (VBT)
- Valence Bond Theory - Orbital Overlap Concept
- Valence Bond Theory - Directional Properties of Bonds
- Valence Bond Theory - Overlapping of Atomic Orbitals
- Valence Bond Theory - Types of Overlapping and Nature of Covalent Bonds
- Valence Bond Theory - Strength of Sigma (σ) bond and pi (π) bond
- Hybridisation
- Hybridisation
- Types of Hybridisation
- Hybridisation of Elements Involving d Orbitals
- Molecular Orbital Theory
- Molecular Orbital Theory
- Formation of Molecular Orbitals - Linear Combination of Atomic Orbitals (LCAO)
- Conditions for the Combination of Atomic Orbitals
- Types of Molecular Orbitals
- Energy Level Diagram for Molecular Orbitals
- Electronic Configuration and Molecular Behaviour
- Bonding in Some Homonuclear Diatomic Molecules
- Hydrogen Bonding
- Hydrogen Bonding
- Cause of Formation of Hydrogen Bond
- Types of Hydrogen Bonding
- Chemical Bonding and Molecular Structure Numericals
- Electronegativity
- Shapes of Simple Molecules
- The Covalent Bond
- Elementary Idea of Metallic Bonding
- Co-ordinate Bonding
