English Medium
Academic Year: 2024-2025
Date & Time: 20th February 2025, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections - A, B, C, D and E.
- Section A - Question Nos. 1 to 20 are multiple choice type questions. Each question carries 1 mark.
- Section B - Question Nos. 21 to 26 are very short answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C - Question Nos. 27 to 33 are short answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D - Question Nos. 34 to 36 are long answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E - Question Nos. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
To get an image of magnification −1 on a screen using a lens of focal length 20 cm, the object distance must be ______.
Less than 20 cm
30 cm
40 cm
80 cm
Chapter:
When a pure-tall pea plant is crossed with a pure-dwarf pea plant, the percentage of tall pea plants in F1 and F2 generation pea plants will be, respectively, ______.
100%; 25%
100%; 50%
100%; 75%
100%; 100%
Chapter:
Plants like rose and banana have lost the capacity to produce ______.
flowers
buds
seeds
fruits
Chapter:
In which one of the following situations a chemical reaction does not occur?
Milk is left open at room temperature during summer.
Grapes get fermented.
An iron nail is left exposed to humid atmosphere.
Melting on glaciers.
Chapter:
The property by virtue of which a solid material can be drawn into thin wires is called ______.
malleability
ductility
rigidity
resistivity
Chapter:
In order to prepare dry hydrogen chloride gas in a humid atmosphere, the gas produced is passed through a guard tube (drying tube) which contains ______.
Calcium chloride
Calcium oxide
Calcium hydroxide
Calcium carbonate
Chapter:
Select from the following a hydrocarbon having one C-C bond and one C≡C bond:
Benzene
Cyclohexane
Butyne
Propyne
Chapter:
Which one of the following has half the number of chromosomes and half the amount of DNA as compared to the non-reproductive body cells?
Male germ cell
Female germ cell
Zygote
Both, male and female germ cells
Chapter:
The essential element taken up from the soil by the plants to synthesize proteins is ______.
Phosphorus
Nitrogen
Iron
Magnesium
Chapter:
Select true statements about lymph from the following:
- Lymph vessels carry lymph through the body and finally open into larger arteries.
- Lymph contains some amount of plasma, proteins and blood cells.
- Lymph contains some amount of plasma, proteins and red blood cells.
- Lymph vessels carry lymph through the body and finally open into larger veins.
The true statements are:
A and B
B and D
A and C
C and D
Chapter:
Which one of the following gets biomagnified at different levels in a food chain?
Carbon monoxide
CFC’s
DDT
Manure
Chapter:
In the food chains given below. Select the most efficient food chain in terms of energy:
Grass → Grasshopper → Frog → Snake
Plants → Deer → Lion
Plants → Man
Phytoplankton → Zooplankton → Small Fish → Big Fish
Chapter:
An optical device ‘X’ is placed obliquely in the path of a narrow parallel beam of light. If the emergent beam gets displaced laterally, the device ‘X’ is ______.
plane mirror
convex mirror
glass slab
glass prism
Chapter:
A piece of wire of resistance ‘R’ is cut lengthwise into three identical parts. These parts are then connected in parallel. If the equivalent resistance of this combination is R', then the value of `R/(R')` is ______.
`1/9`
`1/3`
3
9
Chapter:
The minimum number of identical bulbs of rating 4 V, 6 W that can work safely with desired brightness when connected in series with a 240 V mains supply is ______.
20
40
60
80
Chapter:
An electric bulb is rated 220 V; 11 W. The resistance of its filament when it glows with a power supply of 220 V is ______.
4400 Ω
440 Ω
400 Ω
20 Ω
Chapter:
Assertion (A): All exothermic reactions are accompanied with evolution of heat and light.
Reason (R): Combination reactions may or may not be exothermic.
Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): When ciliary muscles contract, eye lens becomes thin.
Reason (R): Ciliary muscles control the power of the eye lens.
Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Concentrated nitric acid is diluted by adding water slowly to acid with constant stirring.
Reason (R): Concentrated nitric acid is easily soluble in water.
Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): In reptiles, the temperature at which the fertilized eggs are kept decides the sex of the offsprings.
Reason (R): Sex is not genetically determined in some animals.
Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
While burning a magnesium ribbon in air, list two safety measures which should be followed.
Chapter:
What do you observe when magnesium ribbon is burnt in oxygen?
Chapter:
Draw a ray diagram to show the refraction of a ray of light passing through an equilateral glass prism. Mark the angle through which the emergent ray bends from the direction of the incident ray and also name it.
Chapter:
Name the type of lenses used for the correction of presbyopia defect.
Chapter:
Advertisements
Write the structure of the lenses commonly used for the correction of presbyopia defect giving reason for such designs.
Chapter:
“In the human alimentary canal, the small intestine is designed to absorb the digested food.” Justify this statement.
Chapter:
Pure-tall (TT) pea plants are crossed with pure-dwarf (tt) pea plants. The pea plants obtained in F1 generation are then self pollinated to produce F2 generation.
- What do the plants of F1 generation look-like? Justify your answer.
- What is the ratio of pure-tall plants to pure-dwarf plants in F2 generation?
Chapter:
Show the formation of magnesium chloride by electron transfer. Write the name of the cation and anion present in the compound formed. (Atomic Number of Mg = 12, Cl = 17)
Chapter:
How is zinc extracted from its ore? Name the processes involved in the extraction and write chemical equations for the reactions that occur during these processes.
Chapter:
State the role of an electric fuse, used in series with an electrical appliance.
Chapter:
Why should a fuse with a defined rating not be replaced by one with a larger rating in an electric circuit?
Chapter:
State how decomposers maintain a balance in an ecosystem.
Chapter:
Samples of four metals A, B, C and D were added one by one to the following solutions. The results obtained were tabulated as follows:
| Solution → | Iron Sulphate | Copper Sulphate | Zinc Sulphate | Aluminium Sulphate |
| Metal ↓ | ||||
| A | - | Displacement | No reaction | No reaction |
| B | Displacement | Displacement | Displacement | - |
| C | Displacement | ? | - | No reaction |
| D | No reaction | No reaction | No reaction | No reaction |
Use the table above to answer the following questions about metals A, B, C and D:
- Which is the least reactive metal?
- What would be observed if C is added to a solution of copper sulphate?
- Arrange the metals A, B, C and D in the order of their decreasing reactivity.
Chapter:
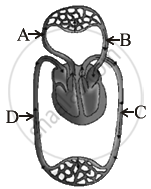
- Study the diagram and name the parts marked as A, B, C and D.
- Write the function of A and C.
Chapter:
The electrical resistivity of three materials A, B and C at 20°C is given below:
| Material | Resistivity (Ω m) |
| A | 1017 |
| B | 44 × 10−6 |
| C | 1.62 × 10−8 |
- Classify these materials as conductor, alloy and insulator.
- Give one example of each of these materials and state one use of each material in the design of an electrical appliance say an electric stove or an electric iron.
Chapter:
If we want to obtain a real and magnified image of an object by using a concave mirror of focal length 18 cm. Where should the object be placed? Use mirror formula to determine the object distance for an image of magnification −2 by this mirror to justify your answer.
Chapter:
Why do we need to balance chemical equations?
Chapter: [1] Chemical Reactions and Equations
Name and state the law that suggests the balancing of a chemical equation.
Chapter:
Balance the following chemical equation:
\[\ce{Zn + H3PO4 -> Zn3(PO4)2 + H2}\]
Chapter:
Give an example of a precipitation reaction and also express the reaction that occurs in the form of a balanced chemical equation.
Chapter:
State two limitations of electrical impulses in multicellular organisms.
Chapter:
Why is chemical communication better than electrical impulses as a means of communication between cells in a multicellular organism?
Chapter:
Give reason why propane has no structural isomers?
Chapter:
Advertisements
Write a balanced equation when butane is burnt in oxygen.
Chapter:
Differentiate between the flames obtained when butane and butyne are both burnt in air in similar conditions.
Chapter:
State the reason why carbon can neither form C4+ cations nor C4− anions but forms covalent compounds.
Chapter: [4] Carbon and its Compounds
What is meant by a functional group in carbon compounds?
Chapter: [4] Carbon and its Compounds
Write in tabular form the structural formula and the functional group present in the following compound:
Ethanol
Chapter:
Write in tabular form the structural formula and the functional group present in the following compound:
Ethanoic acid
Chapter:
Draw the pattern of the magnetic field lines for the two parallel straight conductors carrying current of the same magnitude ‘I’ in opposite directions, as shown. Show the direction of the magnetic field at a point O, which is equidistant from the two conductors. (Consider that the conductors are inserted normal to the plane of a rectangular cardboard.)
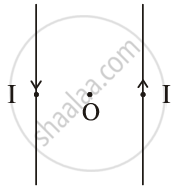
Chapter:
In our houses we receive A.C. electric power of 220 V. In electric iron or electric heater cables having three wires with insulation of three different colours - red, black and green are used to draw current from the mains.
- What are these three different wires called? Name them colourwise.
- What is the potential difference between the red wire and the black wire?
- What is the role of the wire with green insulation in case of accidental leakage of electric current to the metallic body of an electrical appliance?
Chapter:
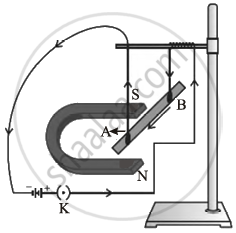
By using the given experimental set-up. How can it be shown that:
- a force is exerted on the current-carrying conductor AB when it is placed in a magnetic field.
- the direction of force can be reversed in two ways.
Chapter:
When will the magnitude of the force be highest?
Chapter:
State Fleming’s left-hand rule.
Chapter: [12] Magnetic Effects of Electric Current
Give one example of an organism that shows a regeneration process.
Chapter:
Give one example of an organism that does not show a regeneration process.
Chapter:
Water in a pond appears dark green and contains filamentous structures. Name these structures and the method by which they reproduce. Explain the process.
Chapter:
Name the part performing the following function in the human male reproductive system:
Carries sperm
Chapter:
Name the part performing the following function in the human male reproductive system:
Production of male gametes.
Chapter:
Name the part performing the following function in the human male reproductive system:
Whose secretion makes the transport of sperms easier.
Chapter:
Name the part performing the following function in the human male reproductive system:
Provide suitable temperature for sperm formation.
Chapter:
Give the side effects caused by the surgical contraceptive procedure.
Chapter:
| The students in a class took a thick sheet of cardboard and made a small hole in its centre. Sunlight was allowed to fall on this small hole and they obtained a narrow beam of white light. A glass prism was taken and this white light was allowed to fall on one of its faces. The prism was turned slowly until the light that comes out of the opposite face of the prism appeared on the nearby screen. They studied this beautiful band of light and concluded that it is a spectrum of white light. |
- Give any one more instance in which this type of spectrum is observed. (1)
- What happens to white light in the above case? (1)
-
- List two conditions necessary to observe a rainbow. (2)
OR - Draw a ray diagram to show the formation of a rainbow. Mark on it points (a), (b) and (c) as given below: (2)
- Where dispersion of light occurs.
- Where light gets reflected internally.
- Where final refraction occurs.
- List two conditions necessary to observe a rainbow. (2)
Chapter:
| Common salt is a very important chemical compound for our daily life. It’s chemical name is sodium chloride and it is used as a raw material in the manufacture of caustic soda, washing soda, baking soda, etc. It is also used in the preservation of pickles, butter, meat, etc. |
- Name the acid and the base from which common salt can be obtained. (1)
- State the nature (acidic/basic/neutral) of sodium chloride. Give a reason for the justification for your answer. (1)
-
- What happens when electric current is passed through an aqueous solution of sodium chloride (called brine)? Name the products obtained along with the corresponding places in the electrolytic cell where each of these products is obtained. (2)
OR - How is washing soda obtained from sodium chloride? Give the chemical equation of the reactions involved in the process. (2)
- What happens when electric current is passed through an aqueous solution of sodium chloride (called brine)? Name the products obtained along with the corresponding places in the electrolytic cell where each of these products is obtained. (2)
Chapter:
| In life there are certain changes in the environment called ‘stimuli’ to which we respond appropriately. Touching a flame suddenly is a dangerous situation for us. One way is to think consciously about the possibility of burning and then moving the hand. But our body has been designed in such a way that we save ourself from such situations immediately. |
- Name the action by which we protect ourself in the situation mentioned above and define it. (1)
- Write the role of (a) motor and (b) relay neuron. (1)
-
- What are the two types of nervous systems in the human body? Name the components of each of them. (2)
OR - Which part of the human brain is responsible for: (2)
- thinking
- picking up a pencil
- controlling blood pressure
- controlling hunger
- What are the two types of nervous systems in the human body? Name the components of each of them. (2)
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2024 - 2025
Previous year Question paper for CBSE Class 10 Science-2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
