Advertisements
Advertisements
प्रश्न
Which of the following does not belong to the same homologous series?
विकल्प
CH4
C2 H6
C3 H8
C4 H8
Advertisements
उत्तर
C4 H8
Explanation -
The succeeding members of the homologues series differ by a —CH2 - unit. The general formula for the alkane is CnH2n+2. Methane (CH4), ethane (C2H6) and propane (C3H8) are members of homologues series for alkanes while butene (C4H8) is the fourth member of homologues series for alkene.
APPEARS IN
संबंधित प्रश्न
State any four characteristics of a homologous series
Write the name and formula of the 2nd member of homologous series having general formula CnH2n + 2.
Write the molecular formula of first two members of homologous series having functional group – OH
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
Give the molecular formula of one homologue of the following:
C3H6
Write the molecular formula of the third member of the homologous series of carbon compounds with general formula CnHO2n+1OH.
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
The molecular formula of the third member of the homologous series of ketones is:
(a) C4H8O
(b) C3H6O
(c) C5H10O
(d) C6H12O
Give the structure of the second member of the alcohol group.
Distinguish between homologous organs and analogous organs. In which category would you place wings of a bird and wings of a bat? Justify your answer giving a suitable reason.
Write names of first four homologous series of alcohols.

The phenomenon in which compounds having different structural formulae have the same molecular formula is called _______.
Write a short note.
Homologous series
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
Observe the structural formula and answer the following questions.
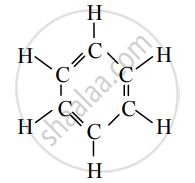
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Complete the following chart by using examples given in brackets.
(isobutylene, cyclohexane, propane, cyclohexene, cyclopentane, benzene, propyne, isobutane, propene)
| Straight chain hydrocarbons | Branched chain hydrocarbons | Cyclic hydrocarbons |
Which of the following pairs can be the successive members of a homologous series?
What is called homologous series? Give any three of its characteristics?
Name the third homologue of alcohols.
