Advertisements
Advertisements
प्रश्न
Using Bohr’s postulates, obtain the expression for the total energy of the electron in the stationary states of the hydrogen atom. Hence draw the energy level diagram showing how the line spectra corresponding to Balmer series occur due to transition between energy levels.
Advertisements
उत्तर
According to Bohr’s postulates, in a hydrogen atom, a single alectron revolves around a nucleus of charge +e. For an electron moving with a uniform speed in a circular orbit os a given radius, the centripetal force is provided by Columb force of attraction between the electron and the nucleus. The gravitational attraction may be neglected as the mass of electron and proton is very small.
So,
`(mv^2)/r = (ke^2)/r^2`
or `mv^2 = (ke^2)/r .............. (1)`
where m = mass of electron
r = radius of electronic orbit
v = velocity of electron.
Again,
`mvr = (nh)/(2π)`
or `v = (nh)/(2πmr)`
From eq.(1), we get,
`m((nh)/(2πmr)^2) = (ke^2)/r`
`=> r = (n^2h^2)/(4π^2kme^2).....................(2)`
(i) Kinetic energy of electron,
`E_k = 1/2 mv^2 = (ke^2)/(2r)`
Using eq (2), we get
`Ek =ke^2/2 (4π^2kme^2)/(n^2h^2)`
=`(4π^2kme^2)/(n^2h^2)`
`(2π^2k^2me^4)/(n^2h^2)`
(ii) Potential energy
`E_p = -(k(e) xx (e))/r = - (ke^2) / r `
Using eq (2), we get
`E^p =-ke^2 xx (4π^2kme^2)/(n^2h^2)`
= `-(4π^2k^2me^4)/(n^2h^2)`
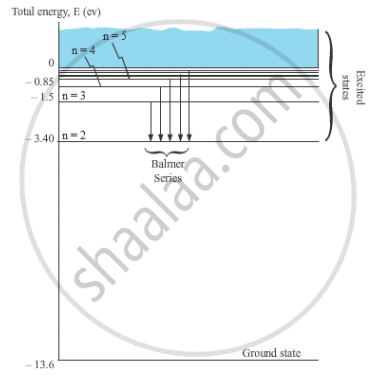
Hence, total energy of the electron in the nth orbit
`E =E_p+E_k =(4π^2k^2me^4)/(n^2h^2)+(2π^2k^2me^4)/(n^2h^2) =- (2π^2k^2me^4)/(n^2h^2) =- (13.6)/n^2 eV `
When the electron in a hydrogen atom jumps from higher energy level to the lower energy level, the difference of energies of the two energy levels is emitted as a radiation of particular wavelength. It is called a spectral line.
In H-atom, when an electron jumps from the orbit ni to orbit nf, the wavelength of the emitted radiation is given by,
`1/λ = R (1/n_f^2 -1/n_i^2)`
Where,
R → Rydberg’s constant = 1.09678 ×107 m−1
For Balmer series, nf = 2 and ni = 3, 4, 5, …
`1/λ = R (1/2^2 -1/n_i^2)`
Where, ni = 3, 4, 5, …
These spectral lines lie in the visible region.
APPEARS IN
संबंधित प्रश्न
State Bohr’s postulate of hydrogen atom which successfully explains the emission lines in the spectrum of hydrogen atom. Use Rydberg formula to determine the wavelength of Hα line. [Given: Rydberg constant R = 1.03 × 107 m−1]
According to Bohr, 'Angular momentum of an orbiting electron is quantized'. What is meant by this statement?
Use Bohr’s model of hydrogen atom to obtain the relationship between the angular momentum and the magnetic moment of the revolving electron.
The ratio of the ionization energy of H and Be+3 is ______.
The binding energy of a H-atom, considering an electron moving around a fixed nuclei (proton), is B = `- (Me^4)/(8n^2ε_0^2h^2)`. (m = electron mass). If one decides to work in a frame of reference where the electron is at rest, the proton would be moving around it. By similar arguments, the binding energy would be
B = `- (Me^4)/(8n^2ε_0^2h^2)` (M = proton mass)
This last expression is not correct because ______.
The Bohr model for the spectra of a H-atom ______.
- will not be applicable to hydrogen in the molecular from.
- will not be applicable as it is for a He-atom.
- is valid only at room temperature.
- predicts continuous as well as discrete spectral lines.
How will the energy of a hydrogen atom change if n increases from 1 to ∞?
The wavelength in Å of the photon that is emitted when an electron in Bohr orbit with n = 2 returns to orbit with n = 1 in H atom is ______ Å. The ionisation potential of the ground state of the H-atom is 2.17 × 10−11 erg.
According to Bohr atom model, in which of the following transitions will the frequency be maximum?
What is meant by ionisation energy?
