Advertisements
Advertisements
प्रश्न
|
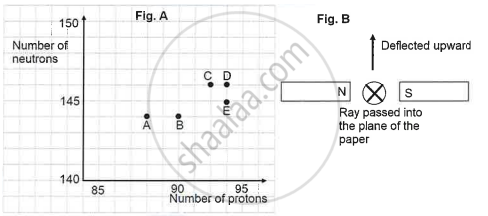
The graph (fig A) illustrates the correlation between the number of protons (x-axis) and the number of neutrons (y-axis) for elements A, B, C, D, and E in the periodic table. These elements are denoted by the letters rather than their conventional symbols. When the element C, depicted in the graph, undergoes radioactive decay, it releases radioactive rays. When these rays are directed into the plane of the paper in the presence of a magnetic field, as indicated in the fig B, they experience deflection, causing them to move upwards.
|
Name the law used to identify the radioactive radiation emitted by the element.
Advertisements
उत्तर
In Figure B, a magnetic field is applied perpendicular to the direction of the radioactive ray. The ray is deflected upward, indicating the type of particle based on the direction of force.
According to Fleming’s Left-Hand Rule, the direction of motion (force) on a charged particle in a magnetic field depends on:
- Thumb → Force (motion)
- Forefinger → Magnetic field (North to South)
- Middle finger → Current (positive charge flow)
Since the particle is deflected upward, and it is moving into the plane of the paper, it must be positively charged, like an alpha (α) particle.
So, Fleming’s Left-Hand Rule helps in identifying the ray as positively charged, i.e., alpha radiation.
APPEARS IN
संबंधित प्रश्न
Which of the following property of a proton can change while it moves freely in a magnetic field? (There may be more than one correct answer.)
State whether the following statement is true or false
The field at the centre of a long circular coil carrying current will be parallel straight lines.
Imagine that you are sitting in a chamber with your back to one wall. An electron beam, moving horizontally from back wall towards the front wall, is deflected by a strong magnetic field to your right side. What is the direction of magnetic field?
State and explain the Clock face rule for determining the polarities of a circular wire carrying current.
The diagram given below represents magnetic field caused by a current-carrying conductor which is:

(a) a long straight wire
(b) a circular coil
(c) a solenoid
(d) a short straight wire
Name three factors on which the magnitude of force on a current carrying conductor placed in a magnetic field depends and state how does the force depend on the factors stated by you.
State whether a magnetic field is associated or not around a moving charge.
Which of the following pattern correctly describes the magnetic field around a long straight wire carrying current?
An alpha particle enters a uniform magnetic field as shown. The direction of force experienced by the alpha particle is ______.

Assertion (A): A current carrying straight conductor experiences a force when placed perpendicular to the direction of magnetic field.
Reason (R): The net charge on a current carrying conductor is always zero.