Advertisements
Advertisements
प्रश्न
The Atomicity of phosphorous is ______
Advertisements
उत्तर
The atomicity of phosphorous is 4.
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
KNO3
Give the name of the element present in the following compound:
Quick lime
Give the name of the element present in the following compound:
Hydrogen bromide
Give the name of the element present in the following compound:
Potassium sulphate
Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur.
Define: Atomic number
In what form do noble gases occur in nature?
Work out the formula for magnesium hydrogencarbonate.
What are (i) ionic compounds, and (ii) molecular compounds ? Give two examples of each type of compounds.
Molecular compounds are usually formed by the combination between :
An element A forms an oxide A2O5.
What is the valency of element A ?
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Give one example of a polyatomic molecule.
Give a reason why –
Ozone is considered triatomic, but phosphorus a tetratomic molecule.
State the atomicity of the following:
Neon
State the atomicity of the following:
Oxygen
State the atomicity of the following:
Argon
Which of the following is a short and scientific way of representing one molecule of an element or compound?
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 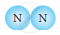 |
|
| 4. | 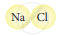 |
If a molecule is made of similar kinds of atoms, then it is called ______ atomic molecule.
Two elements sometimes can form more than one compound.
Give any two examples for heterodiatomic molecules.
Which of the following contains maximum number of molecules?
