Advertisements
Advertisements
प्रश्न
State the atomicity of the following:
Neon
Advertisements
उत्तर
Momoatomic
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
Al2(SO4)3
Write down the name of a compound represented by the following formula:
K2SO4
Write down the name of a compound represented by the following formula:
CaCl2
Write down the name of a compound represented by the following formula:
KNO3
Write down the name of a compound represented by the following formula:
CaCO3
Give the name of the element present in the following compound:
Quick lime
State the term.
The number of atoms present in a molecule of an element _________
Define: Atomic number
Define: Atomic number
Define: Atomic weight :
What is meant by atomicity ? Explain with two examples.
Write the formulae of the following compounds. Also name the elements present in them.
- Water
- Ammonia
- Methane
- Sulphur dioxide
- Ethanol
In what form does oxygen gas occur in nature ?
Work out the formula for magnesium hydrogencarbonate.
An element A forms an oxide A2O5.
What is the valency of element A ?
Calculate the mass of 3.011 × 1024 atoms of carbon.
Give one example of a triatomic molecule
State the main points of the contradiction of Dalton’s atomic theory by the Modern Atomic Theory.
Give a reason why –
Helium is considered monoatomic, but hydrogen a diatomic molecule.
State the atomicity of the following:
Oxygen
State the atomicity of the following:
Argon
Which of the following is a short and scientific way of representing one molecule of an element or compound?
State true of false. If false, give the correct statement.
NaCl represents one molecule of sodium chloride.
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 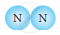 |
|
| 4. | 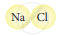 |
If a molecule is made of similar kinds of atoms, then it is called ______ atomic molecule.
Define: Atomicity.
Give any two examples for heterodiatomic molecules.
Which of the following contains maximum number of molecules?
