Advertisements
Advertisements
प्रश्न
State the main points of the contradiction of Dalton’s atomic theory by the Modern Atomic Theory.
Advertisements
उत्तर
Dalton’s Atomic Theory:
The main postulates of the theory are:
- Matter consists of small indivisible particles called – atoms i.e. Atom is the basic unit of matter.
- Atoms of the same element are – alike in all respects i.e. atoms of hydrogen have the same properties as mass, density, and atoms of oxygen are alike in all respects.
- Atoms of different elements are different from each other.
i.e. atoms of hydrogen are different from atoms of oxygen. - Atom can neither be created nor destroyed.
- Atoms combine with other atoms in simple whole-number ratio forming compounds or molecules.
Modern atomic theory contradicted Dalton Atomic Theory as:
- Atom is no longer indivisible as an atom has been divided and has sub-atomic particles
(a) Protons (b) Neutrons (c) Electrons - Atoms of the same element may have different properties
i.e. i.e.11H, 21 H,31H Isotopes - Atoms of different elements may have the same properties
i.e. isobars - Atoms combine with other atoms may not be in the simple whole-number ratio
i.e. C12H22O11(Sugar). - Atom can be destroyed and converted into energy.
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
K2SO4
Give the name of the element present in the following compound:
Quick lime
Give the name of the element present in the following compound:
Baking powder
Define: Atomic number
Define: Atomic weight :
What is meant by atomicity ? Explain with two examples.
In what form does oxygen gas occur in nature ?
In what form do noble gases occur in nature?
What do the following denote ?
- N
- 2N
- N2
- 2N2
One of the forms of a naturally occurring solid compound P is usually used for making the floors of houses. On adding a few drops of dilute hydrochloric acid to P, brisk effervescence are produced. When 50 g of reactant P was heated strongly, than 22 g of a gas Q and 28 g of a solid R were produced as products. Gas Q is the same which produced brisk effervescence on adding dilute HCl to P. Gas Q is said to cause global warming whereas solid R is used for white-washing.
- What is (i) solid P (ii) gas Q, and (iii) solid R.
- What is the total mass of Q and R obtained from 50 g of P ?
- How does the total mass of Q and R formed compare with the mass of P taken ?
- What conclusion do you get from the comparison of masses of products and reactant ?
- Which law of chemical combination is illustrated by the example given in this problem ?
Work out the formula for magnesium hydrogencarbonate.
Molecular compounds are usually formed by the combination between :
An element A forms an oxide A2O5.
What is the valency of element A ?
Calculate the mass of 3.011 × 1024 atoms of carbon.
Correct the following statement:
A molecule of sulphur is monoatomic.
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Give a reason why –
Helium is considered monoatomic, but hydrogen a diatomic molecule.
State the atomicity of the following:
Oxygen
State the atomicity of the following:
Chlorine
State the atomicity of the following:
Sulphur
State true of false. If false, give the correct statement.
NaCl represents one molecule of sodium chloride.
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 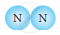 |
|
| 4. | 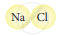 |
Which of the following is a triatomic molecule?
Give any two examples for heterodiatomic molecules.
Derive the relationship between Relative molecular mass and Vapour density.
