Advertisements
Advertisements
प्रश्न
Derive the relationship between Relative molecular mass and Vapour density.
Advertisements
उत्तर
(i) The Relative Molecular Mass of a gas or vapour is the ratio between the mass of one molecule of the gas or vapour to the mass of one atom of Hydrogen.
(ii) Vapour density is the ratio of the mass of a certain volume of a gas or vapour, to the mass of an equal volume of hydrogen, measured under the same conditions of temperature and pressure.
Vapour Density (V.D.) = `"Mass of a given volume of gas or vapour at S.T.P"/"Mass of the same volume of hydrogen"`.
(iii) According to Avogadro’s law, equal volumes of all gases contain an equal number of molecules. Thus, let the number of molecules in one volume = n, then
(iv) V.D at STP = `" Mass of molecules of a gas or vapour at S.T.P"/"Mass of 'n' ‘molecules of hydrogen"`
Cancelling ‘n’ which is common, you get
V.D = `"Mass of 1 molecule of a gas or vapour at S.T.P."/"Mass of 1 molecule of hydrogen"`.
(v) Since hydrogen is diatomic
V.D. = `"Mass of 1 molecule of a gas or vapour at S.T.P."/"Mass of 2 atoms of hydrogen"`
(vi) By comparing the definition of relative molecular mass and vapour density we can write as follows.
V.D. = `"Mass of 1 molecule of a gas or vapour at S.T.P."/(2 × "Mass of 1 atom of hydrogen")`
Relative molecular mass (hydrogen scale) = `"Mass of 1 molecule of a gas or vapour at STP."/"Mass of 1 atom of hydrogen"`
(vii) By substituting the relative molecular mass value in vapour density definition, we get
Vapour density (V.D.) = Relative molecular mass/2
⇒ 2 × vapour density = Relative molecular mass of a gas.
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
K2SO4
Write down the name of a compound represented by the following formula:
KNO3
Give the name of the element present in the following compound:
Potassium sulphate
Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur.
Define: Atomic number
Define: Mass number :
Define: Atomic weight :
What is meant by atomicity ? Explain with two examples.
What do the following denote ?
- N
- 2N
- N2
- 2N2
One of the forms of a naturally occurring solid compound P is usually used for making the floors of houses. On adding a few drops of dilute hydrochloric acid to P, brisk effervescence are produced. When 50 g of reactant P was heated strongly, than 22 g of a gas Q and 28 g of a solid R were produced as products. Gas Q is the same which produced brisk effervescence on adding dilute HCl to P. Gas Q is said to cause global warming whereas solid R is used for white-washing.
- What is (i) solid P (ii) gas Q, and (iii) solid R.
- What is the total mass of Q and R obtained from 50 g of P ?
- How does the total mass of Q and R formed compare with the mass of P taken ?
- What conclusion do you get from the comparison of masses of products and reactant ?
- Which law of chemical combination is illustrated by the example given in this problem ?
An element X forms the following compounds with hydrogen, carbon and oxygen :
H2X, CX2, XO2, XO3
State the three valencies of element X which are illustrated by these compounds.
Correct the following statement:
A molecule of sulphur is monoatomic.
Why are the symbols and formulae of substances important?
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Give one example of a triatomic molecule
Give a reason why –
Helium is considered monoatomic, but hydrogen a diatomic molecule.
Give a reason why –
Ozone is considered triatomic, but phosphorus a tetratomic molecule.
State the atomicity of the following:
Oxygen
State true of false. If false, give the correct statement.
NaCl represents one molecule of sodium chloride.
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 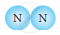 |
|
| 4. | 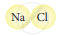 |
If a molecule is made of similar kinds of atoms, then it is called ______ atomic molecule.
The Atomicity of phosphorous is ______
Which of the following contains maximum number of molecules?
Write any two classifications of a molecule.
