Advertisements
Advertisements
प्रश्न
non-stoichiometric defects?
Advertisements
उत्तर
The defect shown by NaCl in non-stoichiometric defect is metal excess defect due to anionic vacancies which is known as F-centre.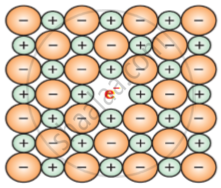
APPEARS IN
संबंधित प्रश्न
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Defects in solids can be studied using
Frenkel defects are not found in alkali metal halides because ____________.
Which is not correct about the Schottky defects?
Alkali halids do not show Frenkel defect because ____________.
What is the effect of Frenkel defect on the density of ionic solids?
In a Schottky defect ____________.
Cations are present in the interstitial sites in ______.
Which of the following point defects are shown by AgBr(s) crystals?
- Schottky defect
- Frenkel defect
- Metal excess defect
- Metal deficiency defect
Which of the following statements are not true?
(i) Vacancy defect results in a decrease in the density of the substance.
(ii) Interstitial defects results in an increase in the density of the substance.
(iii) Impurity defect has no effect on the density of the substance.
(iv) Frankel defect results in an increase in the density of the substance.
