Advertisements
Advertisements
प्रश्न
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
विकल्प
A - (5), B - (4), C - (1), D - (2)
A - (3), B - (4), C - (5), D - (1)
A - (4), B - (3), C - (2), D - (1)
A - (3), B - (4), C - (1), D - (2)
Advertisements
उत्तर
A - (5), B - (4), C - (1), D - (2)
Explanation:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 5. magnesium |
| B. Blood pigment | 4. iron |
| C. Wilkinson catalyst | 1. rhodium |
| D. Vitamin B12 | 2. cobalt |
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
IUPAC name of the following compound is
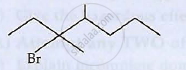
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write IUPAC names of the following compounds

How is Benzonitrile converted to Benzophenone?
Write structures of compounds A and B of the following reaction :

Ligand (en) is an example of ___________.
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
The coordination number of the central ion may be obtained from:
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Which of the following species cannot act as a ligand? Give reason.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
Explain the ligand.
Explain the following, giving two examples:
Heteroleptic
What is meant by didentate ligand?
