Advertisements
Advertisements
प्रश्न
If 4 g of \[\ce{NaOH}\] dissolves in 36 g of \[\ce{H2O}\], calculate the mole fraction of each component in the solution. Also, determine the molarity of solution (specific gravity of solution is 1g mL–1).
Advertisements
उत्तर
Molar mass of \[\ce{NaOH}\] = 40 g/mole
Molar mass of water = 18 g/ mole
Mass of \[\ce{NaOH}\] = 4 g
Number of moles of NaOH = `(4 g)/(40 g)` = 0.1 mol
Number of moles of H2O = `(36 g)/(18 g)` = 2 mol
Mole fraction of water = `(26 g)/("Number of moles of NaOH" + "Number of moles of" H_2O)`
Mole fraction of NaOH = `2/(0.1 + 2) = 2/2.1` = 0.95
Number of moles of `2/0.1 = 0.1/2.1` = 0.047
Number of moles of \[\ce{NaOH}\]
Mass of solution = Mass of solute + Mass of solvent
Mass of \[\ce{NaOH}\] + Mass of water = 4 g + 36 g = 40 g
Specific gravity of solution = 1 g/ml
1 litre = 1000 ml volume of solution = 40 ml
∴ 40 ml = 0.04 litre
Molarity = `"Number of moles of solute"/"Volume in litre" = (0.1 "mol")/(0.04 "litre")` = 2.5 M
APPEARS IN
संबंधित प्रश्न
Calculate the amount of carbon dioxide that could be produced when 1 mole of carbon is burnt in 16 g of dioxygen.
What is the concentration of sugar (C12H22O11) in mol L–1 if its 20 g are dissolved in enough water to make a final volume up to 2 L?
If the density of methanol is 0.793 kg L–1, what is its volume needed for making 2.5 L of its 0.25 M solution?
To make a saturated solution, 36 g of sodium chloride is dissolved in 100 g of water at 293 K. Find its concentration at this temperature.
Solve the following problem:
Write the following number in ordinary decimal form:
43.71 × 10−4
Solve the following problem:
Write the following number in ordinary decimal form:
0.00477 × 105
Perform each of the following calculations. Round off your answers to three digits.
(3.26104) (1.54106)
Perform the following calculation. Round off your answer to three digits.
(8.39 107) (4.53 109)
Solve the following problem:
Perform the following calculation. Round off your answer to three digits.
`(8.94xx10^6)/(4.35xx10^4)`
A 1.000 mL sample of acetone, a common solvent used as a paint remover, was placed in a small bottle whose mass was known to be 38.0015 g.
The following values were obtained when the acetone-filled bottle was weighed: 38.7798 g, 38.7795 g and 38.7801 g. How would you characterise the precision and accuracy of these measurements if the actual mass of the acetone was 0.7791 g?
Solve the following problem:
What weight of calcium oxide will be formed on heating 19.3 g of calcium carbonate?
(At. wt.: Ca = 40; C = 12; O = 16)
When light is passed through water containing a few drops of milk, it shows a bluish tinge. This is due to the ______ of light by milk and the phenomenon is called ______. This indicates that milk is a ______ solution.
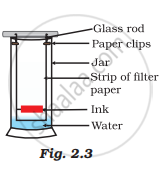
A child wanted to separate the mixture of dyes constituting a sample of ink. He marked a line by the ink on the filter paper and placed the filter paper in a glass containing water as shown in Fig.2.3. The filter paper was removed when the water moved near the top of the filter paper.
(i) What would you expect to see, if the ink contains three different coloured components?
(ii) Name the technique used by the child.
(iii) Suggest one more application of this technique.
If 500 mL of a 5 M solution is diluted to 1500 mL, what will be the molarity of the solution obtained?
What is the difference between molality and molarity?
An aqueous KCl solution of density 1.20 g mL-1 has a molality of 3.30 mol kg-1. The molarity of the solution in mol L-1 is ______. (Nearest integer)
With increase of temperature, which of these changes?
What quantity (in mL) of a 45% acid solution of a monoprotic strong acid must be mixed with a 20% solution of the same acid to produce 800 mL of a 29.875% acid solution?
250 g solution of D-glucose in water contains 10.8% of carbon by weight. The molality of the solution is nearest to ______.
(Given: Atomic weights are H, 1u; C, 12u; O, 16u)
Molarity of liquid HCl will be if the density of the solution is 1.17 g/cc.
